Chemical Peels 101
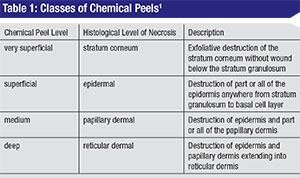
Chemical peeling is the process of applying chemicals to the skin to destroy the outer, damaged layers, thus accelerating the normal process of exfoliation. The ultimate goals of a chemical peel are to reduce skin dyschromias, eliminate fine rhytids, lighten dark circles and reduce actinic changes.1 Surgeons have to be careful when approaching peels in the periocular area, as skin thickness in that region can be as little as 0.2 mm. Eyelid skin differs from other skin in that it lacks subcutaneous fat and has a thinner epidermal-dermal junction and a thin but dense dermis.2 Therefore, it’s difficult to extrapolate the effect of chemical peels on the eyelids based on their effect on other skin areas. Chemical peels penetrating much deeper than the superficial dermis are probably best avoided in order to reduce the risk of scarring and cicatricial ectropion. The depth of penetration depends on acid concentration and the timing of application, and is categorized based on histological depth of necrosis.2 (See Table 1)
 |
Types of Peels
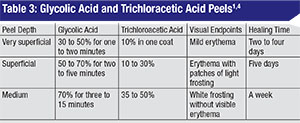
There are a multitude of chemical peels available commercially. Two well-studied products that can be titrated to produce superficial to moderate-depth peels, and are safe for Fitzpatrick I to IV skin types (See Table 2),3 include glycolic acid (GA) and trichloroacetic acid (TCA). (See Tables 3 and 4) These products can be combined with botulinum toxin and fillers in order to obtain an overall improvement in wrinkles, skin tone, texture and clarity. Here are their characteristics and risks:
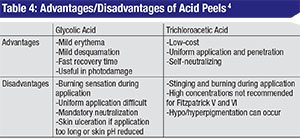
• Glycolic acid. GA is an alpha hydroxy acid (AHA) found naturally in sugar cane. Other AHAs include citric (from citrus fruit), malic (from apples), tartaric (from grapes) and lactic acid (from milk). It’s commercially available in concentrations ranging from 20 to 70% and does require neutralization with an alkaline solution such as 10% sodium bicarbonate solution or water. The gel formulation has a slower penetration time and is easier to control during application.4 GAs are well tolerated and systemically nontoxic and produce superficial to moderate-depth peels capable of significant effects with few complications.1 When applied to skin for three to seven minutes, GA can cause epidermolysis and desquamation of epidermal cells. One double-blind, split-face, vehicle-controlled study showed that four weekly treatments of 50% GA improved skin texture, actinic keratoses and fine wrinkling on clinical exam and demonstrated thinning of the stratum corneum, thickening of the epidermal granular layer and increased prominence of collagen bundles on histologic exam.5
• Trichloroacetic acid. TCA is a self-limited peel that does not require neutralization, and which has proven to be a relatively safe and effective agent for the periocular region.3 The concentration of TCA is most predictive of the depth of peel: 10 to 25% for intra-epidermal effect and 30 to 40% for penetration to the papillary dermis.1 A histopathological study of varying concentrations of TCA applied to eyelid skin in a layered fashion (initial application by cotton-tipped applicator with five minutes of drying time, followed by a second application) demonstrated that even sequential 50% TCA applications failed to penetrate deeper than the papillary dermis.1 Theoretically, the acid must penetrate to the deep reticular layer of the dermis for scarring and contracture to occur.1
• Risks to consider. It’s well worth remembering that increased peel depth, higher acid concentration and longer exposure times increase the risk of scarring and post-inflammatory pigment changes. For GA, experts recommend starting with a lower concentration for the initial peel and gradually increasing the strength over subsequent sessions.6
Minor side effects during and immediately after the peel include erythema, stinging sensation, pulling sensation, mild burning, scaling and transient post-inflammatory hyperpigmentation.5,6
Patient Selection
It’s important to have realistic expectations. Superficial to medium-depth peels produce the best results with mild facial rhytids and minimal dyschromias.3 Superficial peels are effective for freckles, epidermal melasma (blotchy facial pigmentation) and epidermal hyperpigmentation; moderate-depth peels can reduce senile lentigines (small brown patches). Nevi, dermal and mixed melasma, dermal post-inflammatory hyperpigmentation and seborrheic keratoses respond poorly to superficial and medium-depth peels.1 If you need to treat deep rhytids and excessive skin, you should combine a chemical peel with traditional plastic surgery.
 |
 |
Contraindications to chemical peeling include poor healing due to open lesions, radiation, diabetes and photosensitizing medications (e.g., doxycycline, exogenous estrogens). Use of isotretinoin (Accutane) is a strict contraindication for chemical peels for at least one year.1,3 Oral contraceptives may predispose a patient to hyperpigmentation and should be stopped, if possible, in the peri-peel period.1 Be sure to pretreat patients with any history of herpes simplex with oral antiviral medications starting one day prior to the chemical peel and until re-epithelialization is complete.1,3
Preparation
Be sure to obtain photos and informed consent prior to performing any chemical peel procedure. Pretreatment of the skin with retinoic acid preparations (which will be covered in more detail in part two of this series) may allow for better penetration of the chemical peel and faster recovery times. However, this may not be necessary in the periocular area unless the skin is unusually sebaceous or hyperkeratotic.3
Tell the patient to clean the intended treatment area thoroughly with a facial cleanser or non-residue soap the day before the peel, and to avoid makeup or moisturizers. Immediately prior to the peel, clean the patient’s face with acetone or isopropyl alcohol to ensure that any trace of dirt, makeup or oil is removed.2,3
 |
First, position the patient’s head so that it’s tilted at 45 degrees to prevent pooling of the acid.1 Apply a protective layer of petrolatum ointment or zinc oxide paste to the lateral canthus, nasolabial fold, oral commisures and alar groove—all areas that are particularly prone to acid pooling. A cotton-tip applicator can be used to apply gel solutions; a fan-tip brush may be more appropriate for liquid formulations.3 The peel should be applied evenly and quickly with a neutralizing agent readily available. If performing a full-face peel, start with areas of thicker skin first, such as the forehead, and then proceed sequentially to the cheeks, nose, chin, and, finally, the perioral and periocular regions to avoid over-treating areas of thinnest skin.1,3 Skin should be placed on stretch for an even coat, and application should extend through the brow and a little beyond the hair- and jaw-line to avoid clear areas of demarcation.1
A fan directed at the treatment area may relieve mild stinging and burning.
Post-peel Care
The treated areas of skin should be kept moist with a petrolatum-based product. Patients should be counseled to avoid sun exposure, heavy exercise and sweating immediately after a peel.1 They should also avoid picking at the peeling skin, as this can predispose the underlying skin to increased risk of infection, persistent erythema, hyperpigmentation, and scarring.1 A medium-depth peel will cause periocular edema, erythema, crusting and pigmentation for approximately one week after the procedure.1 There’s no need for oral antibiotics in superficial to medium-depth peels unless there is evidence of infection. Once re-epithelialization has occurred, skincare regimens, sunscreens and makeup can be restarted. To minimize risks of post-inflammatory hyperpigmentation, the patient should avoid sun exposure for at least six weeks post-peel.3 Peels should not be repeated any sooner than two weeks, but may be repeated as often as desired as long as you notice evidence of additional benefit.1,6 REVIEW
Dr. Teri Kleinberg practices comprehensive ophthalmology and oculoplastics at Worcester Ophthalmology Associates in Central Massachusetts. She can be reached at 25 Oak Ave., Worcester, MA 01605; Tel: (508) 521-2010, e-mail: terikleinberg@gmail.com.
1. Clark E, Scerri L. Superficial and medium-depth chemical peels. Clinics in Dermatology 2008;26:2:209-218.
2. Dailey RA, et al. Histopathologic changes of the eyelid skin following trichloroacetic acid chemical peel. Ophthalmic Plastic & Reconstructive Surgery 1998;14:1:9-12.
3. Fitzpatrick TB. The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol 1988;124:6:869.
4. Fabbrocini G, De Padova MP, Tosti A. Chemical peels: What’s new and what isn’t new but still works well. Facial plastic surgery: FPS 2009;25:5:329-336.
5. Newman N, Newman A, et al. Clinical improvement of photoaged skin with 50% glycolic acid. A double-blind vehicle-controlled study. Dermatologic surgery 1996;22:5:455-460.
6. Sharad J. Glycolic acid peel therapy–a current review. Clinical, Cosmetic and Investigational Dermatology 2013;6:281.



