Large-screen, “heads-up” 3-D displays for ophthalmic surgery are not a new idea, but this technology initially had some limitations that led many anterior segment surgeons to avoid it. Because many of the drawbacks that were deal-breakers for anterior segment surgeons didn’t apply as much to retinal surgery, some retinal surgeons adopted the technology. Now, proponents say the technology has evolved, and most of those problems have been minimized or eliminated. As a consequence, some anterior segment surgeons are giving this technology a second look.
Here, surgeons discuss their experience using this type of display, explain how it’s changed, and offer some thoughts about why the digital basis of this tech might allow it to help surgeons in novel ways.
An Optical Upgrade
Steve Charles, MD, FACS, FICS, founder of the Charles Retina Institute in Memphis Tennessee, and one of the world’s leading vitreoretinal surgeons, says he’s used Alcon’s Ngenuity system for all of his cases since it was introduced to the market. (Sony and Zeiss also make heads-up 3-D viewing systems; Sony’s is made in partnership with Haag-Streit.)
Dr. Charles notes that early versions of this technology had some significant image latency. “The onscreen visual was noticeably delayed relative to the surgeon’s movements,” he says. “Today the image quality is significantly better; the dynamic range has increased and the latency has been completely eliminated. As a result, interest among both retinal and anterior segment surgeons has increased.”
Rom Kandavel, MD, a partner at the Colvard-Kandavel Eye Center in Encino, California, and volunteer clinical professor at Jules Stein Eye Institute in Los Angeles, recently got his first chance to try using a 3-D heads-up display to perform anterior segment surgery. “At first this technology was presented as a way to improve the ergonomics of surgery,” he says. “That’s a legitimate point. But once I got to try it, I was most impressed by how much better the view is, in almost every way.”
Surgeons note several specific things about the optics that they say are better than a traditional surgical microscope:
• Significantly better depth-of-field. “Not only do you get great clarity, but you get it at almost every focal distance,” says Dr. Kandavel. “You can be in focus at the posterior capsule, the anterior capsule and the endothelium all at the same time. The surgeon can accommodate through those levels, and they’re all in very good focus.”
He points out that the optics of a traditional microscope limit the depth-of-field. “The interpupillary distance and the working distance result in limited stereopsis,” he explains. “It’s a fraction of the stereopsis you get looking at a 55-inch OLED screen from a distance of four feet. With this new technology you can see minute differences in anterior and posterior locations. That’s really important for the everyday cataract surgeon, and it’s also important for residents; it lets them accurately gauge depth while grooving the nucleus, and it helps them detect the posterior capsule once they begin removing pieces.” Dr. Charles agrees, noting that the Ngenuity’s stereo pair of single-chip cameras provide a depth-of-field 4.5 times greater than what a conventional optical operating microscope provides.
Mike Colvard, MD, FACS, director of the aforementioned Colvard-Kandavel Eye Center, and a clinical professor of ophthalmology at the University of California Los Angeles School of Medicine, recently had his first chance to observe this technology being used for anterior segment surgery. “The depth-of-field was astounding,” he reports. “You’re able to look at the cornea, the anterior chamber, the anterior surface of the lens and the posterior aspect of the lens, all at the same time.”
Dr. Kandavel believes retina specialists initially adopted this technology more readily than anterior segment surgeons because of that increased depth-of-field. “It’s particularly helpful for macular hole repair,” he notes. “The digital processing of the image also allows the highlighting of certain structures, which is helpful when peeling membranes, so it caught on in retina first. Nevertheless, the current version of this technology holds a lot of promise for anterior segment surgery.”
• Very high magnification with no loss of clarity. “With typical microscopes, when you start cranking up the magnification, you often lose some clarity and depth-of-field,” Dr. Colvard points out. “You don’t with this technology. When you increase the magnification, you keep the depth-of-field and the clarity.”
Dr. Charles agrees, noting that this is part of the purpose of the large display. “However, when using this system at high magnification, being at the correct viewing distance is crucial,” he points out. “The 55-inch OLED display should be positioned four feet from the surgeon. If you use a smaller display placed closer to the surgeon, the result is vergence-accommodation conflict. Also, the surgeon should wear glasses that have a full distance correction +0.5 D, to optimize the perceived image.”
Dr. Kandavel notes that the improved resolution at high magnification is particularly useful when performing many MIGS procedures. “If you zoom in on the trabecular meshwork, the resolution and focus is excellent,” he says. “You need that visual clarity to work effectively in that space.”
• Clarity out to the edge of the image. “When viewing through a conventional operating microscope, the resolution decreases—and aberrations increase—the further you look from the viewing axis,” notes Dr. Charles. “In contrast, with this technology the cameras are matched to the point-spread function of the optical system. That makes the image resolution for the surgeon just as good at the periphery of the image as it is in the center.”
• Less phototoxicity. “Because the image is digital, the microscope can be at much lower illumination while the screen displays the image at a higher level of brightness,” Dr. Kandavel points out. “That’s not too important for short cataract cases, but it certainly makes a difference in retina cases and longer anterior segment cases.”
• Individual structures can be digitally enhanced. Dr. Kandavel notes that this is another advantage that comes from working with a digital image. “The image can be digitally processed to enhance the contrast or brightness of any structure you choose,” he says.
 |
Non-optical Advantages
The fact that the image appears on a large screen may bring other benefits to this technology as well:
• Better ergonomics for the surgeon. Dr. Kandavel says this is a serious advantage that shouldn’t be overlooked. “Multiple surgeons working in our surgery center have had surgery on their cervical spine because of the strain placed on their backs during surgery,” he notes. “This system can help keep that from happening, especially over many years of performing surgery. You can essentially sit in whatever position you desire and still have a really good view. Physicians always put the patient first; this is a technology that will help the physician.”
He adds that this ergonomic change can be especially helpful when performing some MIGS procedures. “Right now when performing MIGS procedures we generally have to tilt the patient’s head and put on a gonioscope in order to achieve the right angle to see the trabecular meshwork,” he says. “When I use the Ngenuity system, I put the gonioscope on and tilt the scope, but I don’t have to tilt it as much, and it doesn’t affect my head position because I’m just looking at a screen.”
• The surgeon can interact with the surroundings. Dr. Kandavel says this consequence of the heads-up display caught him by surprise. “I underestimated how much more interactive it makes you with your operating room,” he says. “With a microscope, your peripheral vision is almost completely taken up by the oculars. With the heads-up display, you can see your scrub tech, anesthesiologist, even the patient’s body, all while you’re looking at the screen. The whole room is within your visual grasp.”
• Additional information can be added to the screen. “For example, you can do a split screen with the ORA aberrometer,” says Dr. Kandavel. “Or, you can put all of your phaco parameters on the screen, so you can monitor your vacuum, aspiration and energy while performing the surgery. Then you don’t have to turn your head to look at another screen.”
• It has a short learning curve. “I was surprised how quickly people take to the technology, even young surgeons,” says Dr. Colvard. “I watched third-year residents using this technology for the first time at the VA hospital; they had no trouble performing beautifully. So it’s something that people can adapt to very quickly. They weren’t struggling at all. Also, everybody in the room has the same view that the surgeon has—and in 3-D.”
Drawbacks
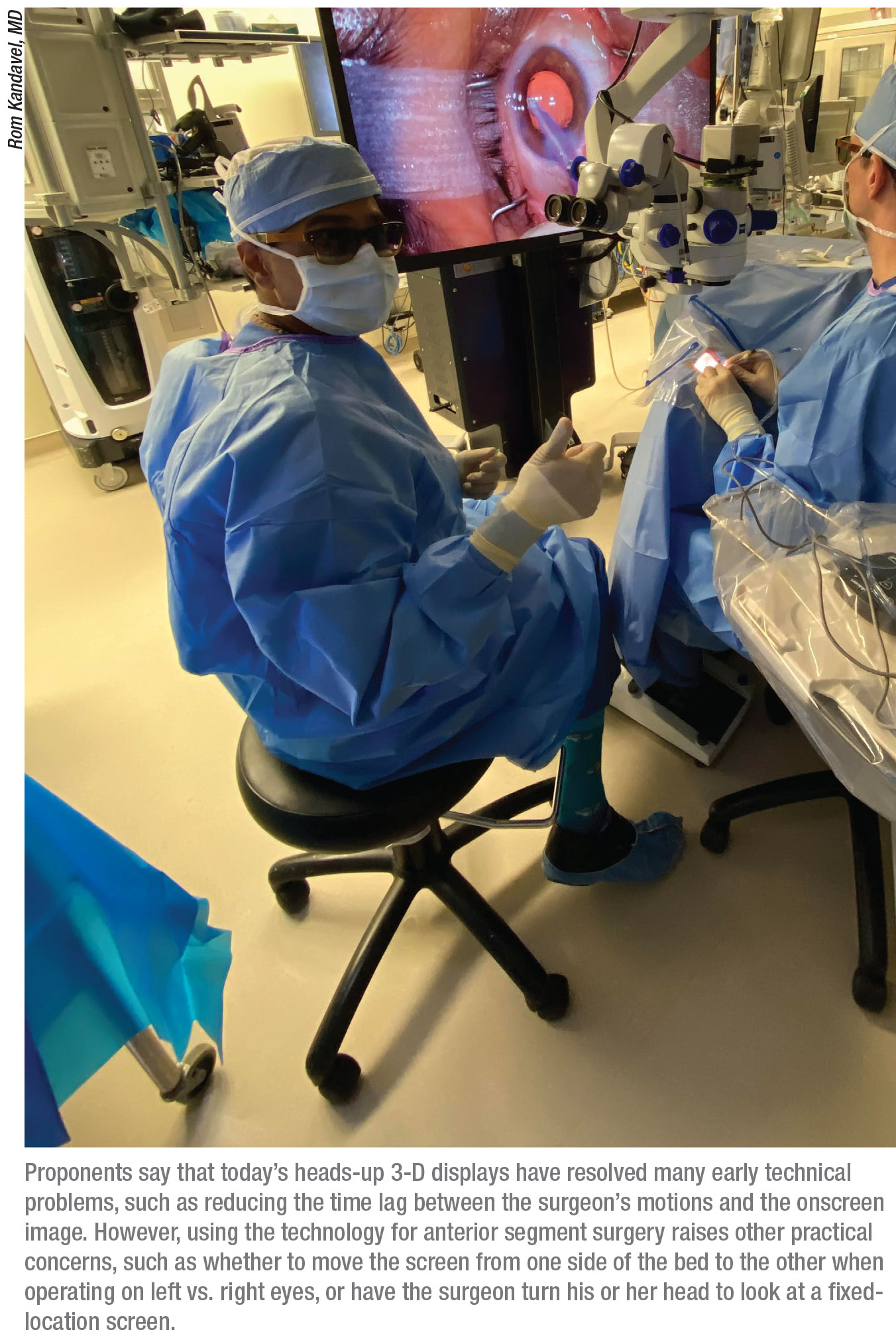
Dr. Kandavel points out a few disadvantages that come with switching to this viewing system. Most of them have to do with managing a large screen in the operating room.
“A retina surgeon operates at the top of the bed, so the screen can be put at the patient’s feet,” he says. “As a result, both the screen and the retina surgeon are in the same position for each case. In contrast, anterior segment surgeons operate left and right, so in order to keep the same view you have to move the screen from one side of the patient to the other—at least in theory. In terms of room turnover and efficiency, that could potentially slow you down.
“However, there are a couple of ways around that,” he continues. “You could schedule all left-eye surgeries in a row, then all right eyes. In that scenario you’d only have to move the screen once, or perhaps a few times a day. It’s also possible to operate with your hands in front of you while looking to your left or right; that way the screen can remain at the patient’s feet, or at the top of the bed. I had some success doing that, but it can be challenging.
“There’s an optimal viewing angle, meaning that you want to be right in front of the screen,” he explains. “Some ovalization of the image and distortion occurs if you’re at a very oblique angle to the screen. This sometimes becomes an issue with an attending resident, because the resident will have the optimal view. The assisting surgeon is accustomed to sitting to the right or left of the resident at the apex of the patient’s head, but with this technology, you want to be located behind your resident so you have the same view the resident has. Otherwise, you’re looking at the screen obliquely, and you may not have the same depth perception they have. That can make it harder to help the resident during surgery.”
In regards to the time lag between the surgeon’s movements and the visual on the screen that initially discouraged many surgeons from adopting this technology, Dr. Kandavel says the time lag has been greatly reduced. “It’s perceptible, but just barely,” he says. “In reality, we don’t generally move at fast enough speeds to make it an issue. Also, after a few cases, you neuroadapt to the slight delay and get used to it. I’ve done multiple cases in a row and had no problem at all.”
 |
There’s also the issue of cost; this technology can cost a practice in the range of $90,000. Surgeons may be reluctant to lay out money for a new viewing system when they feel that their microscopes are already doing a fine job. However, Dr. Kandavel argues that a surgeon can make the numbers work if he or she is in the market for a new microscope.
“Regarding the cost, it appears to be similar to upgrading to a newer microscope,” he says. “In my mind, just getting a newer microscope is an incremental improvement, and it costs about the same as moving to this entirely different way of seeing the surgical field. With this system you’re getting so much more. Instead of making a small step forward, you’re making a giant leap.” (Dr. Kandavel acknowledges that the cost will vary depending on which 3-D system you choose to purchase.)
What Lies Ahead
Dr. Kandavel sees the potential for a number of useful future developments. “For instance, because it’s a digital system, you can ‘lock onto’ structures such as the limbus or the iris,” he says. “You wouldn’t need a microscope pedal because the instrument would automatically remain focused on the area of your choice, regardless of patient movement. It would stay in the center of your field of vision at all times.
“Other even more sophisticated advances should be possible,” he continues. “At some point the software may be able to identify the density of an individual cataract piece in the anterior chamber in real time, based on how the light is passing through it. Then, it could interface with the phaco unit and modulate your energy, vacuum and aspiration accordingly. It could reduce surge and phaco power, all automatically. Or, it could alert you when you’re getting very close to the posterior capsule. And, if the posterior capsule was suddenly sucked up to the aspiration tip—God forbid—the software would immediately release it, faster than a human being could respond. All of this is possible because the system is working with a digital signal. Right now we’re just scratching the surface.”
Dr. Colvard agrees. “One of the old surgical adages is: You can only fix what you can see,” he says. “The joke was, if the doctor can’t see very well, chances are the patient won’t see very well either.
“Being able to see clearly—with great depth-of-field—is a tremendous asset,” he adds. “That’s particularly true with a procedure like capsulorhexis, and especially for a young surgeon.” REVIEW
Dr. Charles is consultant for Alcon but receives no compensation relating to this product. Drs. Colvard and Kandavel report no financial ties to any company or device discussed in this article.



