AMD Inroads
Researchers in the VIEW-1 study of VEGF Trap-Eye for neovascular AMD, sponsored by Regeneron Pharmaceuticals and Bayer Health Care, have one-year follow-up data from their study.
The researchers randomized a total of 1,217 patients from the United States to VTE 0.5 mg monthly (four weeks; 0.5q4wk), 2 mg monthly (2q4wk), 2 mg every two months (eight weeks; 2q8wk) following three monthly doses, or ranibizumab 0.5 mg monthly (Rq4wk). The primary endpoint was the proportion of patients maintaining vision (fewer than 15 ETDRS letters lost) from baseline to week 52.
The researchers randomized a total of 1,217 patients from the United States to VTE 0.5 mg monthly (four weeks; 0.5q4wk), 2 mg monthly (2q4wk), 2 mg every two months (eight weeks; 2q8wk) following three monthly doses, or ranibizumab 0.5 mg monthly (Rq4wk). The primary endpoint was the proportion of patients maintaining vision (fewer than 15 ETDRS letters lost) from baseline to week 52.
At week 52, the percentages of patients maintaining vision in the various groups were: 94 percent (Rq4wk); 95.9 percent (0.5q4wk); 95.1 percent (2q4wk); and 95.1 percent (2q8wk). All study arms were non-inferior to ranibizumab. The mean improvement in best-corrected vision for the groups were: 8.1 letters (Rq4wk); 6.9 letters (0.5q4wk); 10.9 letters (2q4wk) and 7.9 letters (2q8wk). The differences between the study groups and ranibizumab were non-significant in terms of BCVA.
The most frequent emergent adverse events were associated with the injection procedure, the underlying disease and/or the aging process, say the investigators. The most frequent events were conjunctival hemorrhage, macular degeneration, eye pain, vitreous detachment and vitreous floaters.
The researchers say that based upon these results, VEGF Trap-Eye may provide convenient management of wet AMD with predictable dosing every two months.3073
A study of VTE was also carried out internationally in 1,240 patients by the VIEW-2 study, using the same treatment protocols as VIEW-1.
In VIEW-2 at one year, the percentage of patients maintaining vision in the various arms was: 94 percent (Rq4wk); 96.3 percent (0.5q4wk); 95.6 percent (2q4wk); and 95.6 percent (2q8wk). All study arms were non-inferior to ranibizumab. The mean improvement in best-corrected vision for the groups was: 9.4 letters (Rq4wk); 9.7 letters (0.5q4wk); 7.6 letters (2q4wk) and 8.9 letters (2q8wk). The differences between the study groups and ranibizumab were non-significant in terms of BCVA, the researchers say. The most frequent events were conjunctival hemorrhage, retinal hemorrhage and reduced visual acuity.1650
Researchers from the United Kingdom found that a healthy lifestyle may be able to stave off neovascular AMD, even in patients genetically predisposed toward the disease.
In the study, researchers recruited 249 individuals with an angiographically confirmed diagnosis of wet AMD from a macular clinic. Participants were genotyped for both ARMS2 and CFH risk alleles. Covariates available for analysis included age, gender, smoking, body mass index and cardiovascular status. The age at onset was defined as the first record of central visual loss in either eye and was determined from a retrospective examination of clinic notes; this couldn’t be reliably determined in 55 patients, leaving 194 available for analysis.
Linear regression showed that age at onset was approximately three years earlier in those classified as obese compared to those with normal BMI, four years earlier in current smokers when compared to those who had never smoked and five years earlier in those with two copies of the ARMS2 risk allele compared to none. The number of CFH risk alleles wasn’t associated with age at onset. The investigators say that reducing body mass index and stopping smoking may delay the onset of wet AMD, even in the presence of high-risk genotypes.1234
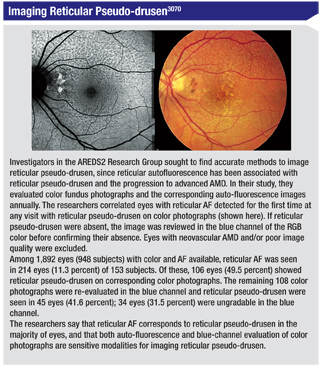 Studies in Occlusion
Studies in Occlusion After a prospective, randomized study, investigators from California say that many patients with central retinal vein occlusion who are treated with ranibizumab will require additional treatments over the long term.
The study consisted of monthly follow-up for three years. In it, physicians randomly assigned patients to receive intravitreal injections of ranibizumab (n=20). After three monthly injections, Group 1 (n=10) received quarterly p.r.n. injections and Group 2 (n=10) received monthly p.r.n. in year one of the study. Beginning in the second year, all subjects were switched to monthly p.r.n. ranibizumab injections at the physician’s discretion for recurrent or persistent macular edema.
Of the initial 20 patients, 19 were included in the combined group analysis to be discussed at ARVO. One patient withdrew from the study after 12 months due to relocation. After one, two and three years of follow-up, the mean BCVA from baseline improved by 1 ±24.8, 7.2 ±21.5, and 6.8 ±23.6 ETDRS letters; mean retinal thickness decreased by 187.8 ±249.75 µm, 292.7 ±241.2 µm and 341.6 ±223.4 µm. The mean number of injections during the various years was: year one, 6.89 ±2.51; year two, 3.42 ±3.86 and year three, 3.05 ±3.20. Fourteen of the 19 patients required additional ranibizumab injections for recurrent macular edema during years two and three of this study. Twelve of 19 patients required injections in year three. The mean number of injections for these subjects during years two and three was 8.78 (median=9, r: one to 21). The mean change in visual acuity for this subgroup (n=14) was an improvement of 1.07 ETDRS letters (median=3, r: -42 to +32) at year three compared to baseline. Five patients didn’t require additional treatment; these subjects received an average of 4.4 injections in the first year with the last injection given at an average of 5.8 months (r: three to 10 months) from baseline. The average change in visual acuity for this subgroup was 22.8 ETDRS letters (median=19.1, r: four to 46) at year three compared to baseline.
No subjects developed iris neovascularization or experienced adverse events related to ranibizumab.
The researchers say that ranibizumab was well-tolerated throughout the entire study, though nearly two-thirds of the patients required additional ranibizumab treatment when followed for three years. The authors say that while many CRVOs will resolve, most patients with macular edema due to CRVO will require continued follow-up and treatment for years.3967
Researchers in an extension of the HORIZON study of ranibizumab for RVO say that a year of treatment with p.r.n. dosing of ranibizumab may not be adequate for patients with CRVO, even though it worked for BRVO patients. The study was sponsored by Genentech, Johns Hopkins University and the Macula Foundation.
 HORIZON (Cohort 2) was an open-label, single-arm, multicenter study, in which patients who completed the 12-month BRAVO and CRUISE trials could receive intravitreal ranibizumab 0.5 mg in intervals of at least 30 days if they had a central subfield thickness
≥250 µm or macular edema that affected visual acuity. The investigators followed the patients for up to two years or until study termination (30 days after Food and Drug Administration approval of ranibizumab for RVO treatment).
HORIZON (Cohort 2) was an open-label, single-arm, multicenter study, in which patients who completed the 12-month BRAVO and CRUISE trials could receive intravitreal ranibizumab 0.5 mg in intervals of at least 30 days if they had a central subfield thickness
≥250 µm or macular edema that affected visual acuity. The investigators followed the patients for up to two years or until study termination (30 days after Food and Drug Administration approval of ranibizumab for RVO treatment).
The researchers enrolled 608 patients in the extension study (304 initially enrolled in BRAVO, 304 initially enrolled in CRUISE). Of these, 205 (67 percent) BRAVO and 181 (60 percent) CRUISE patients completed a year of HORIZON, receiving a mean of 2.5 and 3.8 injections of 0.5-mg ranibizumab, respectively, during those 12 months. BCVA remained stable in BRAVO patients over the first year of HORIZON, with mean changes within ±two letters of baseline vision. In CRUISE patients, however, BCVA declined, with mean reductions of four to five letters from baseline to month 12. Mean central foveal thickness increases from baseline were minimal in BRAVO patients, the researchers say, but ranged from 68 to 88 µm in CRUISE patients at one year. The incidence of serious adverse events across treatment arms during the entire HORIZON trial in the study eye was 2 to 9 percent, and the incidence of serious adverse events potentially related to systemic VEGF inhibition was 1 to 6 percent.4869
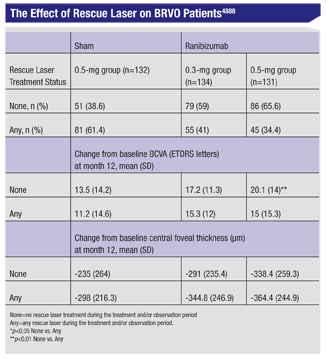
Researchers have also amassed data on the effects of rescue laser treatment in patients in the Genentech-sponsored BRAVO study.
In the study, patients with macular edema following BRVO received six monthly intravitreal injections of 0.3 mg (n=134) or 0.5 mg (n=131) ranibizumab or sham injections (sham/0.5 mg, n=132) for five months (an injection on day zero followed by five monthly injections thereafter) followed by six months of observation (months six to 11), during which eligible patients could receive p.r.n. ranibizumab. Patients could also receive retinal laser one time during the treatment period and once during the observation period if they met visual and anatomic criteria.
At the end of the 12-month study, 61.4 percent of sham/0.5-mg, 41 percent of 0.3-mg, and 34.4 percent of 0.5-mg patients had received at least one retinal laser treatment. Patients in the 0.5-mg group who received laser had a smaller mean change from baseline BCVA compared with those who didn’t receive laser (15 vs. 20.1 letters, p=0.006); 0.3-mg patients who received laser had a longer mean time from diagnosis to day zero than those who didn’t receive laser (5.2 vs. 3.6 months, p=0.026); and 0.5-mg patients who received retinal laser were older, on average, than those who didn’t receive retinal laser (70.5 vs. 65.9 years, p=0.024).
The researchers say that, on average, patients with ME following BRVO who required and received retinal laser in the BRAVO study didn’t achieve the BCVA gains that those who didn’t require or receive retinal laser achieved, and there was no consistent association between baseline characteristics and RL treatment.4888
Researchers in the COPERNICUS Phase III study of Regeneron’s VEGF Trap-Eye for macular edema secondary to CRVO say the drug improves vision in many patients. The researchers either work or consult for Regeneron, or have received funding from the company.
In this randomized, double-masked, controlled Phase III study, 114 patients received six monthly injections of 2-mg VEGF Trap-Eye and 73 patients received control sham injections. Visual acuity was measured as a score based on the total number of ETDRS letters read correctly.
The primary endpoint was the proportion of patients who gained at least 15 ETDRS letters from baseline at 24 weeks. A key secondary endpoint was the mean change in best-corrected visual acuity from baseline to 24 weeks.
The researchers say that the primary endpoint was met in this study: 56.1 percent of patients receiving 2-mg VTE monthly gained at least 15 letters of vision from baseline, compared with 12.3 percent of patients receiving sham injections (p<0.0001). Patients receiving VTE gained a mean of 17.3 letters of vision compared with a mean loss of four letters with sham injection (p<0.001). They say that the most common adverse events were those typically associated with intravitreal injections and/or the underlying disease, and that the proportions of patients who experienced serious ocular adverse events were 3.5 percent in the treatment group and 13.5 percent in the sham group. The incidence of non-ocular serious adverse events was generally well-balanced between the treatment and sham groups, investigators say.6643
Swiss researchers say there may be a risk of increased retinal nonperfusion in occlusion patients treated with anti-VEGF injections, based on a comparison to outcomes of the Central Vein Occlusion Study Group.
In a retrospective study, the investigators analyzed the outcomes of 45 eyes of 45 patients treated with intravitreal ranibizumab for CRVO, BRVO and hemi-central vein occlusion. The follow-up was up to a year. Retinal imaging included fluorescence angiography. Nonperfusion was defined as a retinal area of greater than 25 mm2 (about 10 disc diameters) with loss of capillary perfusion seen on FA. Small macular branch occlusions were excluded.
 The researchers included 24 CRVO/HCVO and 21 BRVO in the study. At baseline they found a rate of 33 percent of nonperfusion (the CVOS rate was 25 percent); it was 35 percent at four months and 65 percent at one year. Nineteen percent (vs. 15 percent of the CVOS patients) of the initially perfused patients converted to nonperfusion within four months follow-up. After a year, 57 percent of these initially perfused patients presented with nonperfusion compared to only 34 percent with ischemia after three years in the CVOS. On average, five injections have been applied until nonperfusion was diagnosed after an average of 325 days.
The researchers included 24 CRVO/HCVO and 21 BRVO in the study. At baseline they found a rate of 33 percent of nonperfusion (the CVOS rate was 25 percent); it was 35 percent at four months and 65 percent at one year. Nineteen percent (vs. 15 percent of the CVOS patients) of the initially perfused patients converted to nonperfusion within four months follow-up. After a year, 57 percent of these initially perfused patients presented with nonperfusion compared to only 34 percent with ischemia after three years in the CVOS. On average, five injections have been applied until nonperfusion was diagnosed after an average of 325 days.
The researchers acknowledge it’s difficult to directly compare the two patient populations, but say the higher conversion rate to nonperfusion in patients treated with anti-VEGF therapy was noteworthy. They say that, until further studies can assess these results, physicians should perform close follow-up exams in patients treated with anti-VEGF therapy, including the use of FA.1256
Diabetic Macular Edema
Researchers from the Fluocinolone Acetonide in Diabetic Macular Edema study (FAME) say the drug provides visual and anatomical improvement over the long term.
The FAME study consisted of two identical, randomized, Phase III, double-masked, sham-controlled, multicenter, parallel-group trials evaluating FA intravitreal inserts (Iluvien, pSivida/Alimera Sciences) in DME patients who had received prior macular laser treatment. The researchers randomly assigned 956 patients to either 0.2-µg/day FA (n=376), 0.5-µg/day FA (n=395), or sham control (n=185). The primary endpoint was the proportion of patients with at least a 15-letter gain in best-corrected vision at month 24. Total study duration was 36 months.
The researchers say that significantly more patients in the 0.2-µg/day FA group experienced at least a 15-letter vision gain at two years vs. control (28.7 percent vs. 16.2 percent, p=0.002). This effect was maintained over three years (28.7 percent vs. 18.9 percent, p=0.018).
They also saw rapid and sustained improvement in mean center-point thickness following treatment with the FA inserts, which remained significant at three years, and report significant improvements from baseline in BCVA letter score. A majority of patients achieved these effects with one study treatment (71.4 percent for sham, 74.4 percent for 0.2 µg/day FA and 70.7 for 0.5 µg/day FA).
By three years, 80 percent of phakic patients in the 0.2-µg/day FA and 87.2 percent in the 0.5-µg/day FA groups underwent cataract surgery vs. 27.3 percent of phakic controls. In terms of intraocular pressure issues, 18.4 of patients in the 0.2-µg/day FA group and 22.9 percent of patients in the 0.5-µg/day FA groups experienced an IOP greater than 30 mmHg at any point in the study vs. 4.3 percent of the sham control patients; 4.8 percent of the 0.2- and 8.1 percent of the 0.5-µg/day FA groups required IOP-lowering surgery vs. only 0.5 percent for controls.
Based on the results of the FAME program, the 0.2-µg/day FA dose has been submitted for regulatory approval. The researchers say that the drug insert significantly improves outcomes in DME with a low risk of incisional procedures for increased IOP.6645
Researchers from the FAME study also examined the effects of FA on IOP.
At baseline, mean IOP measurements were similar among the groups (15 to 15.2 mmHg). The physicians observed mean increases from baseline of between 2 to 3 mmHg for the 0.2-µg/day group and 2 to 4 mmHg for the 0.5-µg/day group from baseline through two years. Subjects in the sham group required fewer IOP-lowering medications (14 percent for sham, vs. 34 percent for the 0.2-µg/day and 44 percent for 0.5-µg/day groups). Based on time to first use of IOP-lowering medication, most of these subjects were treated for the first time by month 12. The incidence of any surgical intervention (i.e., trabeculectomy, glaucoma surgery or vitrectomy for elevated IOP) was lower in the sham group than the active groups (1 percent for sham; 4 percent for 0.2 µg/day and 7 percent for 0.5 µg/day). The most common IOP-related procedure in the study eyes was trabeculectomy (3 percent). Less than 2 percent of all subjects underwent other IOP-lowering surgeries. The majority of IOP-related procedures occurred between months 12 and 24. There were no significant changes in the cup/disk ratio in patients who experienced IOP greater than 25 mmHg on two occasions during the first six months of the study.
The researchers say that the FAME studies demonstrate that, although IOP is elevated in a third of subjects treated with low-dose fluocinolone acetonide, nearly all of these patients are adequately treated by IOP-lowering medications or surgery, and that the ocular hypertensive effect of FA inserts is a manageable side effect.556
Researchers in the randomized, controlled Ranibizumab for Diabetic Macular Edema trial (RISE) say the drug gives sustained results long term.
In RISE, a double-masked, sham-controlled, multicenter trial, investigators randomized 377 patients meeting eligibility criteria (including BCVA 20/40 to 20/320 Snellen equivalent and foveal thickness of at least 275 µm on OCT) to receive monthly ranibizumab (0.5 mg or 0.3 mg) or sham injections. They assessed the need for macular rescue laser starting at month three. The primary efficacy outcome was the proportion of patients gaining at least 15 letters in BCVA from baseline at month 24.
At the primary endpoint, 44.8 percent of the 0.3-mg group and 39.2 percent of the 0.5-mg group gained at least 15 letters, vs. 18.1 percent of sham subjects (p<0.01 for both ranibizumab arms). Mean foveal thickness decreased by 250.6 to 253.1 µm in the treatment groups, and 133.6 µm in the sham.
Ocular adverse events were consistent with prior studies of ranibizumab. The rates of arterial thromboembolic events were similar in the sham and 0.5-mg arms (7.3 and 7.9 percent, respectively), and less common in the ranibizumab 0.3-mg group (3.2 percent). Among ATEs, two strokes (1.6 percent) occurred in the sham group, one (0.8 percent) in the 0.3-mg drug group and five (4 percent) in the 0.5-mg group.
The researchers say that the subjects treated monthly with ranibizumab for 24 months experienced early, sustained and statistically significant improvements in BCVA and foveal thickness compared to subjects in the sham group. The overall rates of ATEs were similar to those reported in other studies of laser, ranibizumab and/or triamcinolone in DME.6647
Researchers from the Ranibizumab for Edema of the mAcula in Diabetes (READ2) trial extended the treatment past the trial’s two-year follow-up period in patients who were willing to continue receiving it.
In the READ2 trial, investigators compared ranibizumab injections (group 1) to laser (group 2) and ranibizumab plus laser (group 3) for six months. Patients were seen every two months (groups 1 and 2) or three months (group 3) and received assigned treatment or ranibizumab for foveal thickness greater than 250 µm. This p.r.n. regimen provided substantial benefit in all three groups at month 24, but wasn’t sufficient to eliminate macular edema in many patients.
To determine if more aggressive treatment could provide increased benefit, patients who agreed to continue participation after two years returned every month and received ranibizumab if their foveal thickness was greater than 250 µm. In this extension study, 30 patients from group 1, 24 from group 2 and 25 from group 3 continued after month 24 through to three years.
The mean improvement in BCVA between baseline and three years was 10.3 in group 1, 1.4 in group 2 and 9.5 letters in group 3. At two years, the mean improvement in vision was 7.1 letters in group 1, three letters in group 2, and 7.1 letters in group 3. The percentage of patients who gained at least three lines from baseline was 35.7 percent in group 1, 9.1 percent in group 2 and 25 percent in group 3 at three years, compared to 25 percent, 9.1 percent, and 20.8 percent at two years. With a mean number of injections of 5.5 (G1), 4.6 (G2), and 2.9 (G3), mean foveal thickness was reduced from measurements of 352.4 (G1), 300.7 (G2) and 249.8 µm (G3) at two years to 282.3, 264.7, and 214.5 µm at three years. The percentage of patients with a foveal thickness of no more than 250 µm was 39.3 percent (G1), 68.2 percent (G2) and 87.5 percent (G3) at three years compared to 32.1 percent, 45.4 percent and 66.7 percent in those groups at two years. There were no ocular or systemic adverse events.
The researchers say that a change in protocol allowing p.r.n. ranibizumab treatment every month instead of every two months resulted in a significant reduction in mean foveal thickness and an improvement in BCVA in groups 1 and 3. They say this suggests that many patients with DME require frequent injections of RBZ to optimally control edema and maximize vision.5332
Researchers in the DaVinci study of VEGF Trap-Eye say the agent is showing potential as a treatment for DME. The researchers consult for and/or receive funding from Regeneron.
In the study, researchers randomized 221 patients to one of five treatment regimens: VTE 0.5 mg every four weeks (0.5q4); 2 mg every four weeks (2q4); 2 mg for three initial monthly doses followed by dosing every eight weeks (2q8); 2 mg for three initial monthly doses followed by dosing as-needed (2p.r.n.); or macular laser photocoagulation.
Mean improvements in letters of BCVA in the VTE groups at week 24 were 8.6 (0.5q4), 11.4 (2q4), 8.5 (2q8) and 10.3 (2p.r.n.), vs. 2.5 letters for the laser group (p<0.01 for each VTE group vs. laser). Mean improvements in letters of BCVA in the VTE groups at week 52 were 11 (0.5q4), 13.1 (2q4), 9.7 (2q8) and 12 (2p.r.n.), vs. -1.3 letters for laser (p<0.0001 for each VTE group versus laser). Proportions of patients with gains in BCVA of at least 15 ETDRS letters in the VTE groups were 41 percent (0.5q4), 46 percent (2q4), 24 percent (2q8), and 42 percent (2p.r.n.) vs. 11 percent for laser.
Mean reductions in central retinal thickness in the VTE groups were -165.4 µm (0.5q4), -227.4 µm (2q4), -187.8 µm (2q8), and -180.3 µm (2p.r.n.) vs. -58.4 µm for laser. The investigators say that VTE was generally well tolerated. The most frequent ocular adverse events were conjunctival hemorrhage, eye pain, hyperemia and increased intraocular pressure.
The researchers say that significant gains in BCVA from baseline achieved at week 24 were maintained or improved up to week 52 in all VTE groups, including the group receiving 2-mg VTE every other month.6646
A researcher in the Diabetic Retinopathy Clinical Research Network in Knoxville, Tenn., found that adding medical therapy to laser treatment for diabetic macular edema provides short-term gains in vision but that the benefit doesn’t last over the long term compared to laser treatment alone.
In the study, 345 eyes were randomly assigned to one of three groups: sham (n=123); 0.5-mg ranibizumab at baseline and four weeks (n=113); or 4-mg triamcinolone at baseline and sham at four weeks (n=109). Follow-up was at one, four, 14, 34 and 56 weeks. The treatment was at the discretion of the investigator after the 14-week visit. At the time of randomization, the eyes had a visual acuity of 20/320 or better and center-involved DME receiving focal/grid laser and diabetic retinopathy receiving prompt panretinal photocoagulation.
Mean changes in visual acuity letter score from baseline were significantly better in the ranibizumab (+1 ±11 letters, p<0.001) and triamcinolone (+2 ±11 letters, p<0.001) groups compared with the sham-injection group (-4 ±14 letters) at the 14-week visit. Reduction in mean central subfield thickness mirrored the visual acuity outcomes. These results weren’t maintained by 56 weeks, however. One eye (0.9 percent, 95-percent CI: 0.02 percent to 4.7 percent) developed endophthalmitis after receiving ranibizumab. Cerebrovascular/cardiovascular events through the 56-week visit occurred in 4 percent of the sham, 7 percent of the ranibizumab and 3 percent of the triamcinolone groups.3970
Dr. Regillo is a professor of ophthalmology at Thomas Jefferson University and director of the Clinical Retina Research Unit at the Wills Eye Institute.




