Age-related macular degeneration is a leading cause of irreversible blindness affecting 8 million Americans.1-3 Most patients with AMD have the dry or nonneovascular form. Although less common, the neovascular form of the disease is responsible for nearly 90 percent of those with severe vision loss. This represents a significant health burden.
Choroidal neovascularization, the hallmark of neovascular AMD, can occur near or under the foveal center (juxtafoveal or subfoveal, respectively), severely reducing visual acuity. Laser ablation of CNV can slow vision loss over the long term, but an immediate reduction in visual acuity due to collateral damage of the overlying photoreceptors is the norm.4 To help minimize this damage, photodynamic therapy was developed to treat CNV near the foveal center. It uses a low-energy laser to excite a verteporfin dye (Visudyne), causing destruction of CNV-specific endothelial cells, thrombus formation and subsequent involution of the neovascularization.5 However, the effect of PDT alone has been surpassed by today's standard treatment for neovascular AMD.
Vascular endothelial growth factor, the major stimulus for CNV growth is now the main target of monoclonal antibodies in neovascular AMD therapy. For the first time in the history of neovascular AMD therapy, an average improvement in visual acuity may be achieved using these agents. Cost-utility analysis has shown that one such agent, ranibizumab, almost doubles the improvement in quality of life of AMD patients compared with PDT.6
Anti-VEGF agents such as ranibizumab (Lucentis) and bevacizumab (Avastin) often require intravitreal injections in regular four- to six-week intervals, with repeated exposure to the risk of endophthalmitis, and frequent follow-up visits.7-12 Although AMD patients are getting better treatment than ever before, they must make significant sacrifices in time. Also, the expense to third-party payers is tremendous. As a result, PDT has been revisited as a complementary treatment to the anti-VEGF agents. Anti-VEGF therapy decreases angiogenesis and vascular permeability resulting in suppression of and the eventual resolution of CNV. PDT may cause relatively immediate closure of CNV. By adding PDT to an anti-VEGF regimen, permanent inhibition of CNV may be possible, with a reduction in the number of required injections. This concept of synergy has been the topic of several published papers and has led to the organization of large, multicentered randomized controlled trials, such as Novartis's
Recent Literature
We previously reported our experience with bevacizumab combined with PDT at the Barnes Retina Institute, and others have since reported their experience using this combination.13-16 In our retrospective review, treatment-naive patients received bevacizumab followed by PDT within a one to two week interval. Treatment with the same combination was repeated based upon persistent subretinal fluid (by optical coherence tomography) or leakage on fluorescein angiography. Of the 40 eyes, 26 (65 percent) required only a single initial treatment of combination therapy to maintain lesion inactivity for at least six months. Thirteen eyes (33 percent) required only a single retreatment, and four of these eyes had subfoveal fibrosis. Only one eye (2 percent) required two retreatments with combination therapy. The mean time to retreatment was 110 days. Thirty-three eyes (83 percent) had stabilization of visual acuity, and 29 eyes (73 percent) had an improvement in visual acuity. Thirteen (33 percent) actually gained three or more lines of visual acuity. Across all lesion subtypes, the mean improvement in visual acuity was 1.73 lines. There were 23 eyes with at least 12-month follow-up, and a remarkable 74 percent of this group achieved stabilization of their visual acuity with only a single combination treatment. There were no ocular or systemic complications noted in this series. This suggests a benefit in combining intravitreal anti-VEGF with PDT.
David Misch, MD, of 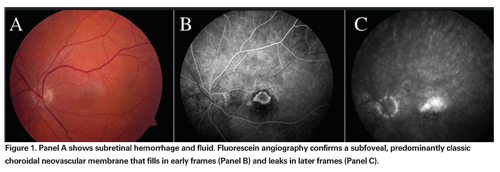
In his second cohort of 44 eyes that had previous therapies, 74 percent achieved stabilization. Twenty-eight percent gained three or more lines of visual acuity, and 41 percent required retreatment.
Another group recently reported its initial results of PDT combined with bevacizumab in 165 eyes.17 Fifty-five were randomized to PDT alone, 55 to 1.25 mg intravitreal bevacizumab alone and 55 to combination therapy. Visual acuity improvement was observed in both the bevacizumab-alone and combination groups. Those in the PDT-alone group experienced a slight worsening of visual acuity. Significant reductions of central foveal thickness measured by OCT were observed in all three groups. The follow-up was three months, and only 23 of the 46 eyes with a visual acuity improvement at one month sustained this improvement at the final follow-up. However, most interesting is the fact that of these 23 eyes, 22 (96 percent) received treatment with bevacizumab combined with PDT, suggesting that PDT helps sustain the visual acuity improvement that is achieved by bevacizumab. Likewise, in another study, 64 percent of 11 patients who previously failed PDT alone required only a single additional treatment session in six months.13 None of the eyes progressed after combination therapy of intravitreal bevacizumab and PDT. The authors of this study also suggest a possible synergistic effect from the combination of intravitreal bevacizumab with verteporfin PDT for the treatment of neovascular AMD.
Other clinicians have added a third agent such as a steroid to their combination therapy. One group treated 17 patients with PDT combined with both intravitreal bevacizumab and triamcinolone.14 The mean central macular thickness was reduced from 395 to 221 µm at 24 weeks of follow-up. Repeat injections of intravitreal bevacizumab alone were performed if fluorescein angiography continued to suggest CNV activity. During an average follow-up period of about four months almost 60 percent required only a single retreatment consisting of bevacizumab alone to maintain initial visual gains and central macular thickness reductions.
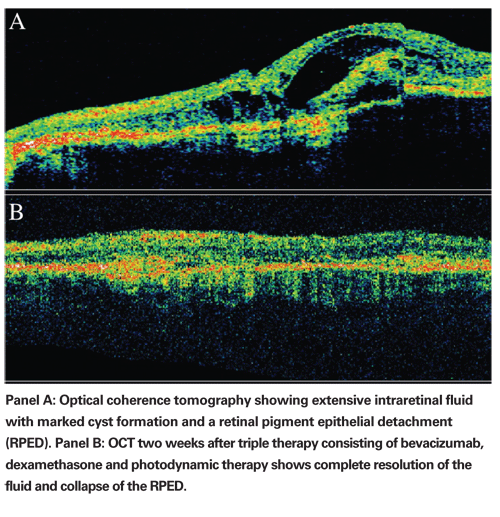
Another study evaluated reduced-fluence PDT in combination with dexamethasone and bevacizumab for choroidal neovascularization secondary to AMD.18 Of 104 patients receiving triple therapy, only five (5 percent) required a second session of triple treatment during the mean follow-up time of 10 months. The triple therapy was complemented in 18 patients (17.3 percent) by an additional intravitreal injection of bevacizumab. The mean increase in visual acuity was 1.8 lines with a mean decrease of 182 µm in retinal thickness. The authors concluded that significant and sustained visual acuity improvement was possible after only one cycle of treatment.
Discussion
These reports suggest a possible benefit in combining PDT with intravitreal bevacizumab. The number of treatments required to stabilize eyes is reduced compared to using anti-VEGF injections alone. At the same time, improvement in visual acuity is achieved. The need for repeated injections with anti-VEGF monotherapy suggests that the neovascular complexes are not extinguished, but lay dormant so long as the neovascular drive from VEGF is quelled. It may be that PDT is necessary to disrupt the architectural components of CNV (accessory support cells) that do not respond to VEGF inhibition. At the very least, it may cause immediate closure of the abnormal vessels. 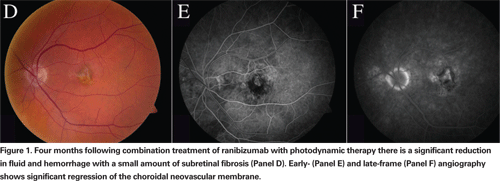
There are, however, many unanswered questions including: the timing of each therapy in relation to one another; timing for retreatment; how to retreat (combination versus monotherapy); and the possible addition of other agents. The fluence at which PDT should be administered when combined with anti-VEGF agents has also been questioned. The VIM (Verteporfin In Minimally Classic CNV) study showed a trend favoring reduced-fluence PDT for minimally classic CNV. We currently treat all cases with reduced fluence to minimize collateral damage. Further, it is not yet known if the optimal final visual outcome is achieved when PDT is used compared to use of intravitreal agents alone. These questions can only be answered through a randomized controlled trial. In the meantime it is important to note that large studies of anti-VEGF agents (MARINA and ANCHOR) exclude eyes with subretinal fibrosis. Many of the studies reviewed here include these eyes, which may limit final visual outcomes, and this must be considered when reviewing these small case series.
Fortunately, the interest in improving patients' vision as well as a desire to use a treatment that is less intrusive into their lives have led to the development of randomized controlled trials to help with these remaining questions.
Neovascular AMD is a chronic disease capable of causing devastating vision loss. Although therapy with anti-VEGF agents is the gold standard, many intravitreal injections are often required. Complementing anti-VEGF therapy with other agents such as PDT may decrease the social and economic burden to patients. Further study is needed to determine at which stage CNV becomes less responsive to VEGF inhibition, and at what critical threshold VEGF sparks de novo or recurrent neovascularization. Ongoing randomized controlled trials will provide many answers to the possible benefit of combining therapies for AMD.
Drs. Smith and Shah are at the Barnes Retina Institute,
The authors have no proprietary interest in this article. Contact Dr. Smith at
1. Age-related Eye Disease Study Research Group. Potential public health impact of Age-Related Eye Disease Study results: AREDS report no. 11. Arch Ophthalmol 2003;121:1621-1624.
2. Eye Diseases Prevalence Research Group. Prevalence of age-related macular degeneration in the
3. Thylefors B. A global initiative for the elimination of avoidable blindness. Am J Ophthalmol 1998;125:90-93.
4. Macular Photocoagulation Study Group: Argon laser photocoagulation for neovascular maculopathy after 5 years: Results from randomized clinical trials. Arch Ophthalmol 1991;109:1109-1114.
5. Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: Two year results of 2 randomized clinical trials - TAP report no. 2. Arch Ophthalmol 2001;119:198-207.
6. Brown MM, Brown GC, Brown H. Value-based medicine and interventions for macular degeneration. Curr Opin Ophthalmol 2007;18:194-200.
7. Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, et al. Ranibizumab for neovascular age-related macular degeneration.
8. Brown DM, Kaiser PK, Michels M, Soubrane G, et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration.
9. Moshfeghi AA, Rosenfeld PJ, Puliafito CA, Michels S, Marcus EN, Lenchus JD, Venkatraman AS. Systemic bevacizumab (Avastin) therapy for neovascular age-related macular degeneration: twenty-four-week results of an uncontrolled open-label clinical study. Ophthalmology 2006;113:2002.
10. Rosenfeld PJ, Moshfeghi AA, Puliafito CA. Optical coherence tomography findings after an intravitreal injection of bevacizumab (Avastin) for neovascular age-related macular degeneration. Ophthalmic Surg Lasers Imaging 2005;36:331-335.
11. Gragoudas ES, Adamis AP, Cunningham ET Jr, et al; VEGF Inhibition Study in Ocular Neovascularization Clinical Trial Group. Pegaptanib for neovascular age-related macular degeneration. N Engl J Med. 2004 Dec 30;351(27):2805-16.
12. Avery RL, Pieramici DJ, Rabena MD, et al. Intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration. Ophthalmology 2006;113: 363–372.e5 [Epub February 3, 2006].
13. Costa RA, Jorge R, Calucci D, Melo LA Jr, Cardillo JA, Scott IU. Intravitreal bevacizumab (Avastin) in combination with verteporfin photodynamic therapy for choroidal neovascularization associated with age-related macular degeneration (IBeVe Study). Graefe's Arch Clin Exp Ophthalmol 2007 Feb 28; [Epub ahead of print]
14. Ahmadieh H, Taei R, Soheilian M, Riazi-Esfahani M, Karkhaneh R, Lashay A, Azarmina M, Dehghan MH, Moradian S. Single-session photodynamic therapy combined with intravitreal bevacizumab and triamcinolone for neovascular age-related macular degeneration. BMC Ophthalmol 2007 Jun 7;7:10.
15. Aggio FB, Melo GB, Höfling-Lima AL, et al. Photodynamic therapy with verteporfin combined with intravitreal injection of bevacizumab for exudative age-related macular degeneration. Acta Ophthalmologica Scandinavica 84 (6), 831–833.
16. Dhalla MS, Shah GK, Blinder KJ, et al. Combined photodynamic therapy with verteporfin and intravitreal bevacizumab for choroidal neovascularization in age-related macular degeneration. Retina 2006 Nov-Dec;26(9):988-93.
17. Lazic R, Gabric N. Verteporfin therapy and intravitreal bevacizumab combined and alone in choroidal neovascularization due to age-related macular degeneration. Ophthalmology 2007;114:1179-85.
18. Augustin AJ, Puls S, Offermann I. Triple therapy for choroidal neovascularization due to age-related macular degeneration: verteporfin PDT, bevacizumab, and dexamethasone. Retina 2007;27:133-40.