The definition of dry eye proffered in 1995 by the National Eye Institute/Industry Workshop on Clinical Trials in Dry Eye1 stated: “Dry eye is a disorder of the tear film due to tear deficiency or excessive tear evaporation, which causes damage to the interpalpebral ocular surface and is associated with symptoms of discomfort.” The first TFOS DEWS report2 updated the definition by—among other things—calling it a disease with multiple underlying causes: “Dry eye is a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance, and tear film instability with potential damage to the ocular surface. It is accompanied by increased osmolarity of the tear film and inflammation of the ocular surface.”
The new definition generated by DEWS II clarifies this even further and incorporates new perspectives supported by recent research: “Dry eye is a multifactorial disease of the ocular surface characterized by a loss of homeostasis of the tear film, and accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles.”3
J. Daniel Nelson, MD, FACS, chair of the DEWS II report, professor of ophthalmology at the University of Minnesota and an ophthalmologist in the HealthPartners Medical Group in St. Paul, explains how the new definition differs from previous definitions—and why it matters.
“All of the definitions of dry-eye disease have been based on the current literature and knowledge at the time,” he says. “The definition suggested by the NIE/Industry workshop defined dry eye as a disorder; in the first DEWS report it was defined as a disease. That’s a key difference. The latter also mentioned that tear film osmolarity and inflammation were involved in the disease. However, the
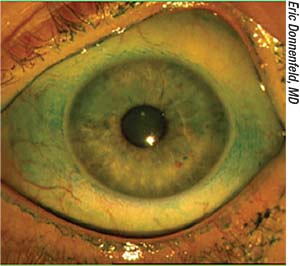 |
| One of the key concepts in the DEWS II definition of dry eye is homeostasis. |
Dr. Nelson says a key choice of wording in the latest definition was the word “homeostasis.” “Homeostasis of the tear film refers to the process of keeping the tear film condition normal, so that it’s able to carry out its normal function—protecting the ocular surface and providing a stable optical interface,” he explains. “Any disturbance or loss of tear film homeostasis can cause symptoms and can lead to, or be due to, tear film instability, hyperosmolarity, ocular surface inflammation and damage, as well as neurosensory abnormalities.
“One reason for using a word like homeostasis is that it keeps the definition from being too narrow,” he continues. “We used to think that the tear film was made up of three layers. Today we know it’s a two-phase tear film with a lipid phase overlying an aqueous-mucin phase composed of a thousand different proteins. If we define DED too specifically, we limit what DED is. If we define it too broadly, then anything could be DED. By defining the disease in terms of a loss of the normal state—homeostasis—the definition avoids those pitfalls. It sets the basis for further research to really define what it is that keeps the tear film in a homeostatic state.”
Dr. Nelson says another key change in the new definition is the mention of neurosensory abnormalities. “One of the issues over the years has been that symptoms often do not correlate with signs,” he says. “A patient may have symptoms, but everything you can measure appears to be normal. That raises the issue of neurosensory abnormalities, which can lead to neuropathic pain. We felt it was important to include that in the definition of the disease.”
Why is a more accurate definition important? Dr. Nelson says one reason is that it affects the ability of clinical trials to provide statistical evidence of a dry-eye product’s efficacy. “We now have two approved drugs for treating dry eye, but many have failed to get approval,” he notes. “One of the main reasons so many have failed is that the definition of dry eye used to select patients for the studies was very broad-based. When you take a group of heterogeneous patients and put them in a study, you’re going to get heterogeneous results. This new definition and classification allows us to be very specific about what we’re testing. We can look for evidence of loss of tear film homeostasis, with specific variables that might include osmolarity, corneal and conjunctival staining and tear film instability. In the past, it was very easy to lump all dry-eye patients together.”
Dr. Nelson says he believes the new definition will help clinicians as well. “A good definition will help you decide whether a patient really does have DED, and that’s going to help determine what treatments you pursue,” he says. “I see many patients who’ve been to two or three different doctors; they come in because they’re not responding to treatment. That’s because right now, most clinicians are basing their diagnosis on symptoms. The new definition says that dry eye is all about the loss of tear film homeostasis. It’s about signs as well as symptoms. If you read the pathophysiology section of the DEWS II report, it talks about the ‘vicious circle.’ This refers to the idea that all four signs—tear film instability, hyperosmolarity, ocular surface inflammation/damage, and neurosensory abnormalities—are like a circle. Any point on that circle can be the starting point and eventually lead to dry-eye disease. So as a clinician diagnosing the problem, you should be looking for signs of hyperosmolarity or inflammation, ocular surface damage, and potentially neurosensory abnormalities. Hopefully the new definition will encourage clinicians to look beyond symptoms.”
1. Lemp MA. Report of the national eye institute/industry workshop on clinical trials in dry eyes. CLAO J 1995;21:4:221e32.
2. The definition and classification of dry eye disease: Report of the definition and classification subcommittee of the international Dry Eye Workshop. Ocul Surf 2007;5:2:75-92.
3. Craig JP, Nichols KK, Akpek EK, et al. TFOS DEWS II definition and classification report. Ocular Surf 2017;15:3:276-83.
Fovista Fails Phase III
Pharmaceutical company Ophthotech announced that the prespecified primary endpoint of mean change in visual acuity at 12 months was not achieved in its Phase III clinical trial undertaken to investigate the potential superiority of Fovista (pegpleranib) anti-platelet-derived growth factor therapy in combination with Eylea (aflibercept) or Avastin (bevacizumab) anti-VEGF therapy, compared with Eylea or Avastin monotherapy for the treatment of wet age-related macular degeneration. The addition of 1.5 mg of Fovista to an Eylea or Avastin regimen didn’t result in benefit, as measured by the mean change in visual acuity at 12 months. REVIEW



