A Manhattan jury last month awarded a former investment banker $7.25 million in damages for vision impairment he claimed resulted from LASIK. The award included $4.5 million in lost income and $2.75 million in pain and suffering, surpassing the record $4 million award to an Arizona airline pilot in 2002.
Mark Schiffer had LASIK surgery in 2000, a week after he first visited an optometrist affiliated with the TLC Laser Eye Center. The surgery was performed by Mark Speaker, MD, then medical director of TLC. Mr. Schiffer's suit claimed he suffered distorted and blurred vision, particularly in his left eye, because the TLC-affiliated doctors failed to determine that he had keratoconus. Mr. Schiffer claimed his vision impairment forced him to leave his highly paid Wall Street career and take a job with his father's Long Island banking security company.
TLC was released from the suit. Dr. Speaker claimed that Mr. Schiffer did not suffer from keratoconus and that his injuries were far less severe than claimed.
AN UNORTHODOX NEW APPROACH TO TREATING POTENTIALLY conditions involving corneal edema has been developed by Daljit Singh, MD, in Amritsar, India. Dr. Singh is the creator of Transciliary Filtration, a glaucoma procedure approved by the Food and Drug Administration in 2004, and is also known for his work supporting the existence of conjunctival and corneal lymphatics. His new procedure may offer hope in situations where a donor cornea is not available.
Much of Dr. Singh's work relies on the Fugo Blade, a handheld instrument that generates a plasma field around a fine filament; the plasma field disintegrates tissue, analogous to the effect of an excimer laser. Unlike electrocautery, no burning is involved, and the Fugo Blade creates no clinically significant collateral tissue damage. As a result, holes created by the Fugo Blade show no scarring and allow fluid in neighboring tissues to drain freely in or out.
At the 2005 American Society of Cataract and Refractive Surgery meeting, Dr. Singh presented early results of his new procedure. The new technique, which involves creating multiple vertical pits in the cornea outside of the visual axis, was developed in an attempt to help an 85-year-old male corneal-decompensation patient who otherwise would have remained blind. The patient had received cataract surgery and iris claw lens implantation in his one functioning eye 18 years earlier. "His eye urgently needed keratoplasty," says Dr. Singh. "Unfortunately, it's extremely difficult to get donor corneas in India, and he had no money to pay a specialist. Conventional options were out of the question."
 |
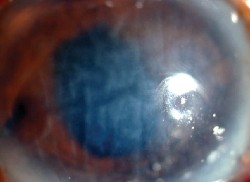 |
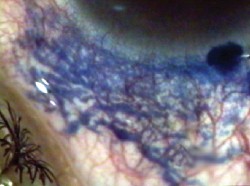
| An injection of trypan blue in the corneal periphery seeps into the lymphatics of the conjunctiva. | This one-eyed patient needed keratoplasty, but no donor corneas were available. |
Dr. Singh describes the technique as follows: "Most eyes with corneal decompensation have a central corneal thickness of 850 µm or more, so I never make corneal pits more than 500 µm deep," he says. "The depth is controlled by selecting a Fugo Blade tip of that length. With surface anesthesia and the eye fixated to prevent movement, the activated filament of the Fugo Blade is repeatedly jabbed vertically into the cornea, producing corneal pits about 200-µm wide. Antibiotics and antifungal meds are instilled for about a month, by which time the tracks are firmly closed on the surface.
"It appears that the corneal pits facilitate the movement of fluid from the deeper layers of the cornea into the anterior layers and then into the conjunctival lymphatics," he says. Dr. Singh emphasizes that the pits must be formed without burning or charring the tissues so fluid can seep into them. "I don't know of any instrument besides the Fugo Blade capable of doing this," he notes.
Twenty-four hours after using this technique for the first time, the patient's eye looked better. "He remained in our clinic for observation for 10 days," says Dr. Singh. "By then he was able to move around on his own, and the eye continued to improve. At this point I let him return home—a trip of a thousand miles. I encouraged him to return for follow-up, but had little hope that he would.
"One and a half months later, to my surprise, he entered my clinic smiling," he continues. "His corrected vision was 20/40." Interestingly, Dr. Singh also notes that the patient's endothelial count had improved from 450/mm2 10 days after surgery to 860/mm2. "He returned again at one year," he adds, "with the same vision and endothelial cell count.
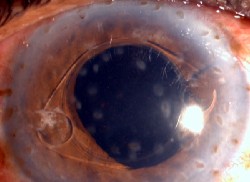 |
| A month and a half after treatment, the patient's corrected vision was 20/40. |
Dr. Singh notes that it can take months to achieve a clear central cornea. "Unfortunately," he says, "follow-up has been very uneven because of the financial and distance constraints facing many of my patients, even though I treat them free of charge if they can't afford to pay."
Dr. Singh adds that while the procedure has been a success so far, a great deal of study needs to be done to understand what really causes the improvement. In the meantime, this new procedure may offer hope to patients when a corneal graft cannot be provided.
For more information, contact Dr. Singh at daljits1@mac.com.
Iowa AG Targets 'See Clearly'
THE IOWA ATTORNEY GENERAL HAS FILED A CONSUMER FRAUD lawsuit against the company that markets kits it claims would naturally improve vision without the need for surgery, according to the Associated Press.
"We allege that the company made dramatic claims for its product that it could not substantiate, including representations that consumers who used the method could quickly and easily free themselves of having to wear glasses or contact lenses," said Tom Miller, at a news conference.
The lawsuit was filed in Polk County District Court against Vision Improvement Technologies Inc.
The lawsuit charges the company marketed a kit called the "See Clearly Method," containing manuals, charts and video and audio tapes demonstrating eye exercises and other techniques it claimed would naturally improve vision.
Mr. Miller said the company has been marketing the kits nationally since 2001, selling the kits on a 30-day trial basis and calling them "risk free." Mr. Miller said many complaints received by his office alleged it was difficult to return the products and get a refund.
The lawsuit claims that 575 Iowans bought the kits between Jan. 1 and Aug. 27, 2004. About half returned the kits.
The lawsuit said the methods were based on similar claims of William H. Bates, who promoted similar ideas in the early 1900s. The lawsuit said those notions have been dismissed by eye care professionals for decades.
LAST MONTH, THE FDA APPROVED ADVANCED MEDICAL OPTICS" Tecnis intraocular lens on an acrylic platform.
"FDA approval of the Tecnis acrylic IOL marks an important milestone for AMO because it represents the first approved product that combines the outstanding optical technology from our 2004 acquisition of the Pfizer ophthalmic surgical business, with our existing advanced IOL materials expertise," said AMO President and CEO Jim Mazzo. "This approval broadens our portfolio of acrylic IOLs, providing more options for our customers and their patients."
The Tecnis lens reduces spherical aberration and improves functional vision in varying light conditions, which is likely to provide a meaningful safety benefit for older drivers and pedestrians. In a simulated night driving study, patients viewing a rural road through the Tecnis lens identified a pedestrian hazard significantly sooner than through a traditional spherical IOL.
The Tecnis also provided a 45-foot advantage in detection and identification distance. At 55 miles per hour, this would provide an additional 0.5 seconds to perceive and react to a pedestrian hazard.
AMO plans to release the Tecnis foldable acrylic IOL commercially in the United States and Europe in September 2005.