Though the majority of cataract surgeries are uneventful and result in excellent outcomes, there's always that occasional case that stresses a surgeon's skill and nerves. Whether you're facing a brunescent cataract or a case of intraoperative floppy iris syndrome, surgeons say it pays to be prepared with the right tools and the proper mindset for the job. In this article, several experts share their tips for dealing with brunescent lenses, IFIS, toxic anterior segment syndrome and zonular instability.
The Brunescent Cataract
Surgeons say these darkened lenses usually demand a change in your technique and your instruments.
"Brunescent cataracts truly are a different beast," says
For these lenses, Dr. Arbisser performs a technique she calls circumferential disassembly. In it, she makes superficial cracks through the periphery of the lens until she can open it in a manner similar to a clamshell. She then goes in and removes the endonucleus to completely debulk the dense part of the lens. "Naturally, you need a nice soft shell of Viscoat [Alcon] against the endothelium and then a cohesive agent to get that in place," she says. "And you need a good hydrodissection so you can rotate the lens without putting stress on the zonules." She then debulks to the nucleified epinucleus, brings the posterior fibers up into the anterior chamber to break them apart and emulsifies the rest of the nucleified endonucleus. "That way, virtually all the energy is used at the iris plane or below," she explains. "The epinucleus is there to constantly protect the capsule that has no good cortical cushion and keeps the equator expanded throughout the removal of the bulk of the lens. This is helpful because these patients often have weak or fragile zonules."
In terms of phaco parameters, Dr. Arbisser uses burst mode with a panel setting that's high enough to achieve her goal of burrowing in and removing material. "I find that because I like to get small pieces and bring them to the center out of the endonucleus, which means using the Kelman tip on its side a little bit and getting the endonucleus out, I need good holding power," she says. "So, I prefer not to use torsional phaco for this particular kind of dense, brunescent lens. Since you're taking a piece out and eating it and so on, with burst mode you're not using continuous energy so you have a very low ultrasound power. Everything's well-controlled. I can take the most brunescent lens and, as long as there's no disastrous zonular pathology, pretty much guarantee 20/20 vision the next morning."
Tucson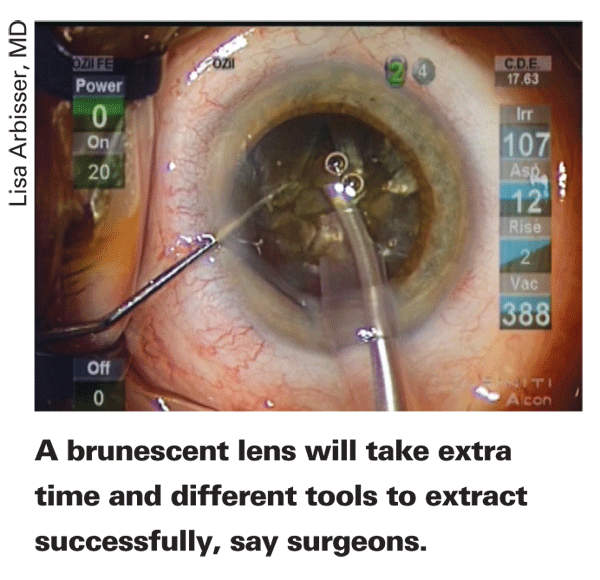
Dr. Fishkind uses a sharp Nichamin quick-chop maneuver to get into the nuclear tissue, making sure to put the chopper very close to the phaco tip, then moves the phaco tip up and right while moving the chopper down and to the left to try to create a quick chop. This only partially cracks the nucleus in some instances, in which case he'll do another chop in the same place, just a little deeper, and try it again. He then frees the phaco tip from the nuclear material, rotates the material a couple of millimeters, and goes back in to do another chop. He makes multiple chops for 360 degrees until he's got the nucleus broken up into 2-mm pie segments. "I don't even try to remove anything," he says, "I just make multiple chops. Eventually, when you get enough of these, the nucleus begins to lose its rigidity. It becomes more flexible and then it becomes easier to try to remove the pie-shaped segments. After I've created enough and I start to see that the nucleus is breakable, I'll break out a segment and pull it up to the iris plane and emulsify there. Once I get the first fragment out, I then have a little more room to work with and I can take the next one, crack it and bring it out the rest of the way.
"You may not be able to crack the back [of the cataract] because it's leathery," Dr. Fishkind continues, "so you may have to pull it apart or get it partially pulled apart and then pull it forward. Then, from the back, you have to snap it against the phaco tip, with no phaco power. You just pull it against the tip and slice it across."
Intraoperative Floppy Iris Syndrome
This appears to be most likely to occur in patients taking tamsulosin (Flomax) for the treatment of benign prostatic hyperplasia, and it can pose a challenge to surgeons who suddenly find themselves dealing with a floppy iris and small pupil. Here are some approaches to dealing with IFIS.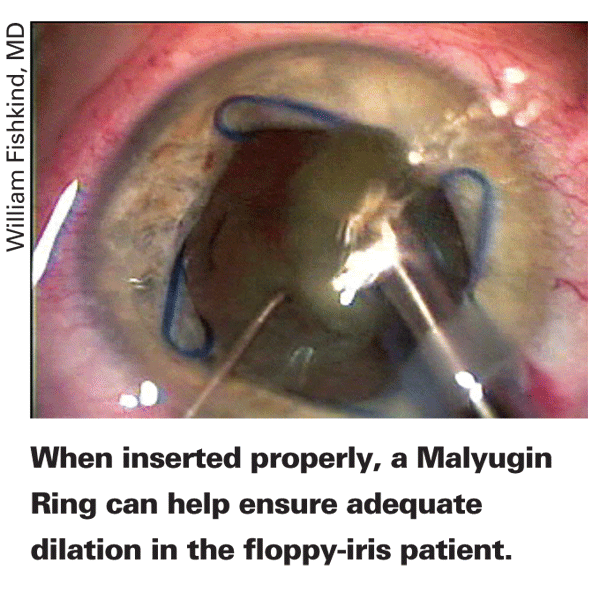
"For tamsulosin patients who dilate at least moderately well, I believe that intracameral epinephrine is of value and is quite safe and easy to use," says
Los Angeles
Dr. Masket says if the initial pharmaceutical approaches and the DisCoVisc fail, he'll protect the cornea with viscoelastic and then administer a small amount of 1:4,000 non-preserved, bisulfite-free epinephrine under the iris.
If the pupil still isn't adequate, many surgeons will use a device. For several surgeons, this takes the form of the Malyugin ring (MST,
"I try to put the ring in the eye with the first little clip opposite the incision and then attempt to engage the pupil with that clip," Dr. Fishkind continues. "Then, I put the left lateral one in, followed by the right lateral and then the inferior one. I do it all with a Kuglen hook. Once it's all in, I center up the pupil with the hook and I'm ready to operate. To remove the ring, I release the subincisional clips first, the right lateral, left lateral and then pull the ring out gently through the incision with the Kuglen hook."
Toxic Anterior Segment Syndrome
TASS is marked by rapid-onset (12 to 48 hours after surgery) severe, non-infectious anterior segment inflammation. If not treated quickly, TASS can lead to serious sequelae, such as corneal edema requiring keratoplasty, severe glaucoma due to trabecular meshwork damage and chronic cystoid macular edema. However, treatment isn't the issue with TASS. Instead, it's the elusive nature of its causes, which can be anything from treatment material in cannulas to preservatives in a medication. The challenge is catching and eliminating these potentially problematic agents or contaminated instruments before they cause inflammation.
Nick Mamalis, MD, of the
To help prevent TASS in your center or hospital OR, Dr. Mamalis offers the following advice:
"First, flush any reusable handpieces that are used in surgery, such as the phaco handpiece and the I/A handpiece," he says. "This means flushing them with 120 cc of sterile water, either deionized or distilled. The problem is these handpieces can have residues of cortex or ophthalmic viscosurgical devices inside them. Though these things don't cause infection, they can be inflammatogenic and cause TASS.
"The second area is the use of cannulas and small-bore cannulas," Dr. Mamalis adds. "The problem with these is it's difficult to clean them adequately once material such as OVD or cortex has dried inside them. They have to be flushed immediately at the conclusion of a case."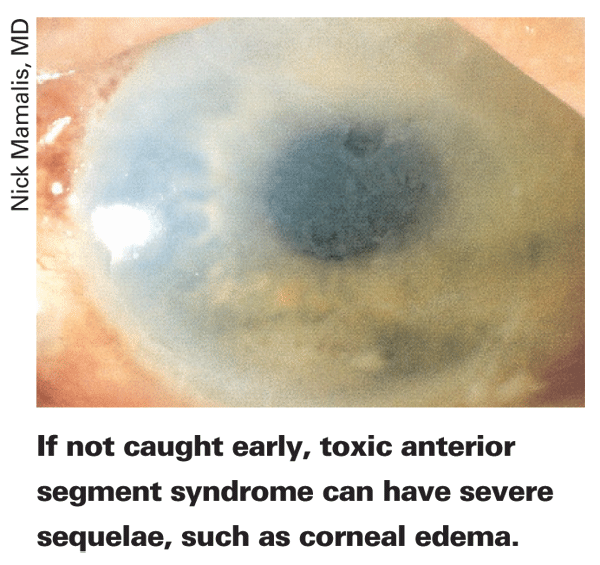
Dr. Mamalis also says there are some issues involved with detergents and enzymes that are sometimes used to clean instruments. "A lot of centers, especially in
Dr. Mamalis says a lot of centers will use an ultrasound bath to shake off debris left on instruments. The problem arises when the bath isn't properly cleaned after each use, which fosters the growth of bacteria. "It's not so much the bacteria that's at issue," says Dr. Mamalis. "Instead, a lot of bacteria, especially the gram-negative organisms, can have an endotoxin that's left on the bacterial wall that's heat-stable. So, when you sterilize instruments after being put in this ultrasound bath, the heat from the sterilization will kill the bacteria, so there's no infection, but the endotoxin can resist the heat sterilization. This endotoxin residue can go into the eye and create TASS. We recommend you drain and clean the bath thoroughly after each use." Also, avoid epinephrine with preservatives or stabilizers and avoid improperly custom-mixed antibiotics.
The latest development with the TASS Task Force is a strengthening of its relationship with the U.S. Food and Drug Administration. "We're now working directly with the FDA to try to be a little more proactive in recognizing potential outbreaks, especially those related to a particular product or medication," says Dr. Mamalis. "We're working with the agency on protocols to try to catch outbreaks in the early stages." If you're currently dealing with an outbreak of TASS, there's a TASS questionnaire located on both the
Zonular Instability
Unlike IFIS, where you might have some warning of a potential problem, zonular instability can occur at any time, so it pays to be ready for it. Here's how an expert in managing loose or absent zonules,
First, Dr. Rosenthal says to look for the obvious, and not-so-obvious, preop signs that you may be in for zonular issues, such as the following:
• pseudoexfoliation;
• a history of trauma;
• a history of collagen vascular disease such as Marfan's;
• previous surgery, especially RK, vitrectomy or filtration surgery;
• phacodenesis;
• tilting of the crystalline lens;
• vitreous slipping out around the crystalline lens; and
• increased retro-iridal space.
However, Dr. Rosenthal says about a fifth of the cases of zonular instability he sees have none of the signs. "You always need to be ready for the treatment of zonular laxity for every case you go to the OR with," avers Dr. Rosenthal. "I think it's important for every OR that does eye surgery to have capsular tension rings available."
Dr. Rosenthal divides zonular disease into two categories: one in which the zonules maintain their normal strength but in which a sector or quadrant is damaged, usually by trauma; and a category where there's diffuse zonular disease, usually from a genetic cause, in which the zonules lose their elastic and tensile properties, and either break or stretch. The management and prognosis differ for each cause.
For patients with the first category of zonular difficulty, damaged zonules, Dr. Rosenthal says in many cases there will be the sign, noted above, of a wisp of vitreous peeking around the cataract through an area of broken zonules. "Sometimes you'll also see a flattening at the edge of the intact capsule instead of a radius of curvature at the slit lamp because there's no tension on the zonule," he says.
In instances of zonular damage, Dr. Rosenthal says to assume that the zonular dehiscence is twice what you see at the slit lamp. If you think it's two clock hours it's probably four, for instance. "The initial management if there's vitreous present beside the lens is a pars plana approach to an anterior vitrectomy, so you're pulling vitreous down into the vitreous cavity," he says. "You want to keep the probe of the vitrector pointed slightly posteriorly or even completely posteriorly to avoid traumatizing the posterior capsule. I like to use a bimanual technique in which infusion is on a separate cannula, usually in the anterior chamber, so the flow is from the anterior chamber to the posterior. In this way, whatever vitreous is in the anterior chamber is being followed by a fluid replenishment with positive pressure as it's being sucked into the posterior chamber, discouraging further vitreous prolapse. The key is to remove as little vitreous as possible, because you don't want to remove the support of the vitreous base." He'll often use Healon V to support the anterior chamber because the capsule is often flaccid in the area of focal zonulysis, which the Healon V will help tamponade, or there are small pupils that can be viscodilated, as in cases of zonular disease due to pseudoexfoliation. The Healon can also plug the area of zonular opening to prevent further vitreous prolapse. He'll also stain the capsule with Trypan blue to help make the capsulorhexis easier.
The next decision involves the potential use of a capsular tension ring, and needs to be made regardless of the cause of the zonular instability. "The capsular tension ring will stabilize the capsular bag by equalizing the distribution of the zonular forces around the entire circumference of the lens," Dr. Rosenthal explains.
When placing the ring, timing is everything. "You want to place the ring as late as possible but as early as you need to," says Dr. Rosenthal. "By that I mean placing the ring at the end of the case is the most technically simple; it avoids the problem of trapping cortex behind the ring and will still stabilize the eye postop, but it has no value for stabilizing the eye during surgery. If there's profound zonular disease, it may be necessary to insert the ring at the beginning of the case or halfway through. The problem there is it can trap cortex against the capsule. My technique to avoid this problem is to remove the anterior and equatorial cortex before doing the nuclear removal, and then place a large amount of Healon V in the anterior part of the anterior capsule, pressing all of the remaining cortex and the nucleus toward the posterior pole and leaving the equator and anterior capsule free of cortex. I then place the capsular tension ring under the anterior rim of the capsule where there's no cortex."
For very profound zonular loss, Dr. Rosenthal will sometimes reinforce his CTR insertion technique with an optic capture/iris suture maneuver. In this technique, he places the lens in the ciliary sulcus and then "retro-places" the optic through the capsulorhexis so the optic is in the bag but the haptics are in the sulcus. He'll then suture the two externalized haptics to the peripheral iris. "So we're stabilizing the capsular bag with the capsular tension ring and, at the same time, stabilizing the haptics, and therefore the IOL, by suturing them to the iris," says Dr. Rosenthal.
Though cataract complexities such as IFIS and zonular instability can be challenging to deal with, surgeons say if you follow a systematic approach and come armed with the right instruments, such complications don't have to result in poor outcomes.