|
Ann P. Murchison, MD |
Anophthalmia is generally an acquired condition; true congenital anophthalmia is very rare. Probably the most common reason for surgical removal of the eye or its contents is blinding trauma; painful blind eyes, prevention of sympathetic ophthalmia, intraocular tumor or endophthalmitis are all common reasons for acquired anophthalmia. While the indication for surgery is usually clear, the choice between enucleation and evisceration is often less clear and more controversial. Given the frequency of enucleation and evisceration, it is important for all ophthalmologists to understand the management of anophthalmic patients and recognize complications in order to optimize patient appearance and comfort. This article reviews the history and discusses the evaluation techniques for the anophthalmic socket.
History
The Chinese possibly first performed enucleation as early as 2600 BC, but the first report was by Johannes Lange in 1555.1 In 1583, Georg Bartisch gave the first report of enucleation with minor changes in technique2 and in 1646 Fabrici de Hilden first reported a peritomy. The procedure advanced to a more modern technique in 1841 when Irish physician O'Ferrall and French physician Bonnet simultaneously reported their technique of extraocular muscle disinsertion.3 Since that advancement the procedure became much more widely used.
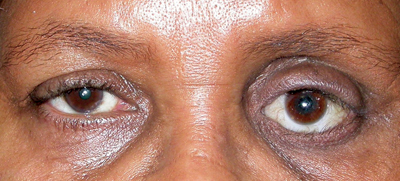 |
| Figure 1. Anophthalmia, OS. Compared to the unaffected right eye, the left eye socket is sunken superiorly and the left lower eyelid is retracted. |
Evisceration seems to have first been performed unintentionally by James Beer in 1817 after an expulsive choroidal hemorrhage; in 1874 J.F. Noyes completed the first planned evisceration. There were several modifications of the procedure, later discarded, until Alfred Graefe and P.H. Mules' more modern version in 1884.2 The first implant reported was a glass sphere for evisceration by Mules in 1885.4 One year later Adams Frost used the implantation in enucleation, and his procedure was refined by Lang. Since that time, many materials and shapes have been tried from glass to metals, ivory to rubber, wool to cartilage and many others.
In recent history the advent of inert plastics has changed the implants used. Today most implants are solid (polymethyl methacrylate or silicone), porous (hydroxyapatite and high-density polyethylene) or autogenous dermis-fat grafts. The most recent published survey in 2003 reported spherical porous polyethylene to be the most commonly used implant.5
Regardless of the implant used and the surgical technique, the goals of the surgeon remain the same; treating the underlying condition, replacing orbital volume, maximizing motility and providing the most comfortable and aesthetically symmetric appearance. All anophthalmic sockets undergo changes including orbital fat atrophy and changes in the orbital circulation. These changes and the placement of an orbital implant can lead to a variety of complications and management difficulties. For these reasons it is important that the general ophthalmologist be able to adequately evaluate and treat simple problems of the anophthalmic socket.
Evaluation
When a patient with anophthalmia presents there are a few basic questions the general ophthalmologist should ask. Does the patient have pain with the prosthetic in? Does the prosthesis fall out? Is there discharge or bleeding from the socket? How old is the current prosthetic and when was the last time it was polished? Does the patient have polycarbonate glasses to protect the seeing eye? Is the patient happy with the cosmesis and movement of the prosthetic?
 |
 |
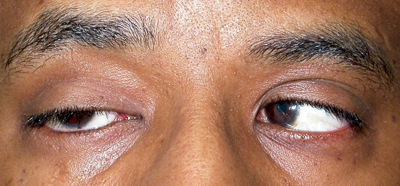 |
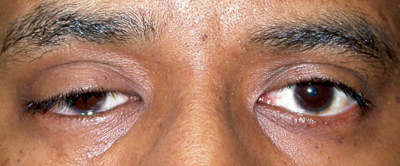
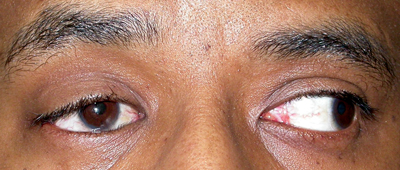
| Figure 2a-c. Anophthalmia, OD. Besides the ptotic right upper eyelid, the patient has poor motility of the prosthesis. The motility should also be observed with the prosthesis out of the socket. If eyesocket motility is better than prosthesis motility, a motility peg or magnetic coupling device, or a better fitting prosthesis may improve motility. Ptosis in an anophthalmic socket can often be improved with a new prosthesis as well. |
In evaluating the anophthalmic socket it is important to look at the general appearance and symmetry compared to the normal side. With the prosthetic in place the patient should be evaluated for enophthalmos or prosthetic malposition. The lower eyelid should be evaluated for laxity. Lash position and lid margin position should be noted, as entropion can indicate socket contracture. The superior sulcus should be checked for deepening and symmetry with the opposite side (See Figure 1). The upper eyelid position should be noted for ptosis, and levator function should be evaluated.
The movement of the prosthetic should be evaluated compared to the seeing eye. Poor movement can be due to fornix abnormalities, enophthalmos or poor prosthetic depth. Any socket or prosthetic abnormality should be addressed, and if the patient is still unhappy with the motility, there are surgical prosthetic-implant coupling methods that may improve motility for the patient (See Figures 2 a to c).
The prosthetic can then be removed and evaluated. If the prosthesis is thick it may be placing pressure on the lower lid and could be camouflaging low orbital volume. Also it should be noted whether the prosthetic is smooth and clean. The socket should be evaluated for inflammation, excessive mucous, giant papillary conjunctivitis under the upper eyelid and pyogenic granulomas. The foniceal depth should be noted, if the superior fornix is excessively deep or if the fornices are not well defined. The tissue over the implant should be examined for thinning, fistula or a defect. Lastly, on palpation of the socket, the presence or absence of an implant and the position of the implant should be noted.
Of course the seeing eye must also have careful exams, with the frequency of exams determined by the patient's age, history and the health of the eye.
Treatments
Enophthalmos, whether determined by prosthetic position or a deep superior sulcus, can be addressed surgically by augmenting the orbital volume. A variety of surgical techniques can be applied, including subperiosteal implants or secondary orbital implants. A discussion of implants and surgical techniques is beyond the scope of this general review. However, for the patient interested in a non-surgical fix, placing a +2 D sphere or higher over the affected side will magnify the eye socket making the enophthalmos less noticeable.
Lower eyelid laxity can be due to in part to a large prosthetic; if so the prosthetic should be remade before any surgical procedure. The commonly performed tarsal strip procedure can correct the laxity; less commonly, lower eyelid sling procedures are at times used.
Entropion must be carefully evaluated to determine if the etiology is due to horizontal laxity or mild socket contracture. Horizontal laxity can be treated as mentioned above. Socket contracture is treated surgically with a variety of techniques, at times including grafting of mucous membrane or other reconstructive material.
Upper eyelid ptosis is common in the anophthalmic socket. This can be a true ptosis or a pseudoptosis due to poor support from the prosthetic or poor orbital volume and implant location. As is well known, any orbital surgery, such as volume augmentation, should precede eyelid correction. Once any socket problems have been corrected the ptosis can be addressed. If the ptosis is mild the ocularist can build up the prosthetic superiorly to support the upper eyelid. However as this increases the weight of the prosthetic it can begin a cycle of problems in the future by inducing lower eyelid laxity, which then leads to a deeper superior sulcus and the need for a larger prosthetic in a perpetuating cycle. If the ptosis is to be addressed surgically the ophthalmologist should keep in mind that levator strength may be underestimated.6
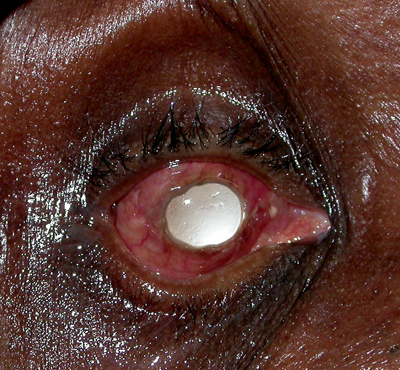 |
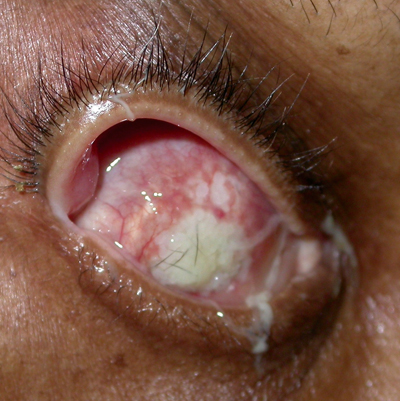 |
| Figure 3a & b. Anophthalmia with exposed implant, OD. In both cases the patient complained of discharge out of the right eye socket. Figure A shows an exposure of a smooth, acrylic implant. In these cases, since the implant is not integrated to the eye socket tissue, the implant is usually replaced. Figure B shows an exposed, porous implant. These implants are integrated with the orbit soft tissue, and, therefore, the defect can often be repaired leaving the implant in place. |
Discharge is a common complaint of the anophthalmic patient, and there can be many underlying causes. The most common etiology is giant papillary conjunctivitis (GPC); this inflammatory reaction is diagnosed by visualizing papillae of 1 mm or greater on the superior tarsal conjunctiva. The conjunctiva becomes edematous and excessive discharge can be seen. As the pathogenesis is a combination of immunologic response to the prosthetic and the mechanical trauma of the prosthetic, one may think the optimal treatment is removal of the prosthetic, as it is with contact lens-related GPC. However, since socket contraction can occur without the prosthetic or a conformer in place, this is usually the last management option after prosthetic polishing and topical cromolyn sodium or topical steroids. Other conditions that may cause discharge are poor prosthetic fit, extruding implant (See Figures 3a and b), pyogenic granuloma in the socket, excessively deep fornices or nasolacrimal duct obstruction.
Other Considerations
Orbital pain in the anophthalmic socket can be difficult to diagnose as the etiology can range from prosthetic irritation or migration/ extrusion of the implant to depression. One should also rule out lacrimal insufficiency, inflammation (scleritis, sympathetic ophthalmia,7 and GPC) and possible recurrent tumors. Amputation neuromas can result in pain with eye movement, but are uncommon unless the socket has undergone significant traction or compression. The amputation neuroma is an exuberant overgrowth of neurons and connective tissue at the transected nerve. Brain and sinus diseases, such as trigeminal neuralgia, meningioma and sinusitis, can also cause referred socket pain and should be considered. Reflex sympathetic dystrophy, a characteristic burning socket pain associated with facial trauma/infection/tumor,8 should also be considered.
Psychogenic factors, such as drug-seeking behavior, can also lead to pain, but these are diagnoses of exclusion. Chronic pain syndromes, which can overlap with psychogenic factors, can also complicate the evaluation and are diagnosed in conjunction with other specialists after ruling out orbital/prosthetic etiologies. In these cases the patients nearly always need referral to an ocularist for assessment and prosthetic modification and/or polishing. If the etiology is not clear and persists after prosthetic polishing and lubrication, the patient may need a CT scan to aid in diagnosis.
It is worth mentioning here that pediatric anophthalmia can be even more difficult to manage, especially as the orbit of a child is not fully developed until after age 5 years. As the soft tissue and orbit can need expansion, these often challenging cases may require multiple surgeries and sometimes subspecialty management to avoid orbital and facial asymmetry.
The anophthalmic socket has a unique set of problems and requires a different clinical and surgical approach than a socket with a globe. The management of these patients should be carried out with close communication between the ophthalmologist and ocularist to achieve optimal comfort and cosmesis for patients.
Drs. Murchison and Bernardino are in Oculoplastics and Orbital Surgery in the Department of Ophthalmology at Emory University School of Medicine. They have no financial interest in the topics/products discussed. Contact Dr. Bernardino at 1365B Clifton Rd., NE, Atlanta, GA 30302. Phone: (404) 778-4144, fax: (404) 778-4415, e-mail: crbernardino@mac.com.
1. Beard CH. Enucleation. In Ophthalmic Surgery. Philadelphia: P. Blakiston's Son & Co. 1910:457-477.
2. Luce CM. A short history of enucleation. Int Ophthalmol Clin 1970;10:678.
3. Fox SA. Enucleation and Allied Procedure. In Ophthalmic Plastic Surgery, 4th Ed. New York: Grune & Stratton, Inc. 1970:475-501.
4. Mules PH: Evisceration of the globe, with artificial vitreous. Trans Ophthalmol Soc UK 1885;5:200.
5. Su GW, Yen MT. Current Trends in Managing the Anophthalmic Socket after Primary Enucleation and Evisceration. Ophthal Plast Reconstr Surg. 2004; 29:279-90.
6. Nunnery WR, Cepela M. Levator function in the evaluation and management of blepharoptosis. Ophthalmol Clin North Am. 1991;4:1-16.
7. Glatt HJ, Googe PB, Powers T, Apple DJ. Anopthalmic Socket Pain. Am J Ophthalmol. 1993;116:357-62.
8. Waldman, SD. Reflex sympathetic dystrophy. Intern Med 11:62, 1991.



