The disproportionality analysis found reported odds ratios for developing macular degeneration of 3.82 (2.94-4.96) for alendronate, 2.40 (1.49-3.86) for ibandronate and 2.87 (1.58-5.19) for risedronate. The case-control analysis, including 6,367 cases of macular degeneration and 63,670 controls, found that subjects regularly using oral bisphosphonates for three years had an adjusted odds ratio of 1.59 (1.38-1.82) for developing wet AMD. The self-controlled case series analysis included 193 cases of macular degeneration on continuous bisphosphonate therapy; it found the rate ratio was 1.22 (95% CI: 0.76-1.95) after one year of bisphosphonate exposure, which rose to 1.87 (95% CI: 1.32-2.67) after five years of exposure. The ratio didn’t differ significantly between men and women.
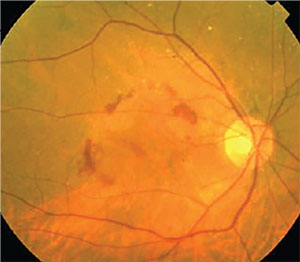 |
“Bisphosphonates are pro-inflammatory drugs that have been shown to cause inflammatory eye conditions such as uveitis and scleritis,” explains study author Mahyar Etminan, PharmD, MSc, assistant professor of ophthalmology and visual sciences at The University of British Columbia. “We hypothesized that these drugs might increase the risk of wet macular degeneration, another inflammatory eye condition. In terms of our study’s limitations, we didn’t have information on body mass index or smoking, and our study needs to be validated by other large epidemiologic studies.”
“This information is interesting and should be verified by randomized clinical trials,” comments David S. Boyer, MD, a retina specialist in Los Angeles and part of the clinical faculty at the Doheny Eye Institute, USC School of Medicine. “Data from excellent long-term studies such as the AREDs studies and population studies such as Blue Mountain, as well as the randomized clinical trials used for approval of the anti-osteoarthritis medications, would also be useful.”
“There are significant limitations to this kind of study,” notes Jason Hsu, MD, assistant director of retina research at Wills Eye Hospital in Philadelphia. “In particular, the authors did not control for potential confounders such as smoking. It’s conceivable that more patients on bisphosphonates may have been smokers, especially given the well-established association between smoking and increased risk of osteoporosis. Smoking also significantly increases the risk of AMD and may have therefore falsely contributed to the appearance that bisphosphonates are to blame. It will be critical that future studies take into account more of these potential confounders before we can draw any conclusions about the association between bisphosphonates and AMD.”
In the meantime, Dr. Etminan believes that doctors prescribing these drugs should perform a risk-benefit analysis. “If a patient is at high risk of future osteoporosis, the benefits of the drugs may outweigh the risks,” he says. “However, the risk of macular degeneration and other adverse events in women who have a low risk of fracture might outweigh its benefits.”
U.S. and India to Collaborate on Glaucoma Research
In a joint effort, researchers from the United States and India have begun to look at the genetic risk factors and traits linked to glaucoma in a study funded by the National Eye Institute and India’s Department of Biotechnology.
This new study will build upon a report from 2010 that looked at the value of genetic mapping to identify traits that are risk factors for diseases such as glaucoma and macular degeneration—age-related diseases that are the leading cause of blindness in many countries, are costly to treat and threaten patients’ ability to live independently. Dr. Ronnie George, senior consultant in the Department of Glaucoma at the Vision Research Foundation, explains the aim of the joint venture: “The goals of the project are to identify genetic markers for glaucoma and factors that influence glaucoma, like intraocular pressure and the optic disc, by targeting families with consanguineous pedigrees,” he says. Earlier studies have suggested that consanguineous pedigrees of sufficient size and structure will provide more significant, quantitative trait mapping than a similarly sized collection of nuclear families.
 |
U.S. researchers will focus on genetic analyses to identify the risk factors that might be associated with glaucoma, an effort that will be headed up by Janey Wiggs, MD, PhD, associate director of the Ocular Genomics Institute at Massachusetts Eye and Ear/Harvard Medical School. Both she and Dr. George emphasize the fact that the study is a collaborative effort and that, without both study arms, the study would not be possible. To accomplish her task of tagging the genetic risk factors, Dr. Wiggs will compare the DNA of those with glaucoma to those without, focusing on their genetic differences in the hopes of identifying some genetic precursor to the disease.
In addition to identifying risk factors for glaucoma, one of the study’s long-term goals is to use this approach to develop and support a genotype/phenotype database that will enable investigators to discover, assess and validate genes and biomarkers responsible for traits that contribute to other complex ocular diseases.
By identifying the genes for disorders such as glaucoma, the investigators hope to get a better idea of these diseases’ underlying causes. Ideally, they say, this knowledge will lead to new methods of diagnosis and treatment. “The results will not be directly applicable to the management of glaucoma,” says Dr. George. “However, analyzing the genes and pathways for factors that influence the disease could help identify potential drug targets.” REVIEW



