Recent years have seen dramatic advances in treating age-related macular degeneration. Now another promising new technology is under examination. The Epi-Rad 90 Ophthalmic System, developed by NeoVista Inc. in
Tony Moses, vice president of marketing and sales at NeoVista, explains that the key element of the Epi-Rad system is a protective handpiece housing a small strand of Strontium-90; the tip of the handpiece is inserted into the eye through a core vitrectomy channel, at which point the isotope is moved down into the tip so the radiation can affect the targeted area.
"Strontium-90 is an isotope that's been used in ophthalmology for many years, so ophthalmologists are familiar with it," explains Mr. Moses. "It has a very high central radiation impact, but minimizes damage to outlying tissues; the level of radiation drops rapidly as you move toward the periphery of the lesion. Also, Strontium-90 only penetrates about 3 mm into tissue. Results so far have shown that this specific radiation and treatment protocol doesn't cause any unwanted problems in the back of the eye."
How Epi-Rad 90 Works
Mr. Moses says the handpiece is similar to a phaco handpiece in size, shape and weight. "The cannula, or tip, is an enclosed, hollow stainless steel 20-ga. tube, a size retinal specialists are accustomed to working with," he notes. "Inside the device is an extrusion wire. On the tip of the wire, inside a miniature housing, is the Strontium-90 isotope."
Mr. Moses explains that the Epi-Rad system includes a "multi-channel tester," used to ensure that the Strontium-90 is correctly positioned inside the cannula before the device is placed into the eye. "The tester reads the radiation coming from two different positions inside the cannula," he says. "If the readings don't fall within the correct specifications and a second test confirms that, the handpiece is deemed nonusable and we replace it."
During the procedure a core vitrectomy is done under local anesthesia to create a channel for the Epi-Rad 90 handpiece. "Once the tip is inside the eye, the surgeon activates the isotope by releasing the lever on the handpiece," says Mr. Moses. "This allows the wire to drop down through the radiation-blocking handpiece into the cannula, placing the Strontium-90 just a little bit back from the tip."
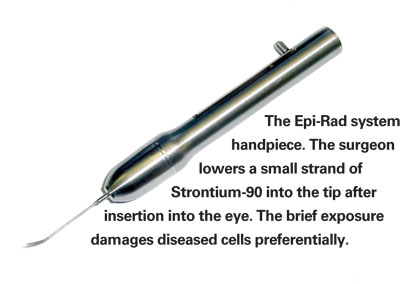
Once the radiation is activated, the surgeon holds the tip in place. "The surgeon knows where to hold the tip to maximize effectiveness because of the training we've provided," Mr. Moses explains. "Also, during the procedure, the surgeon has a previously made fluorescein image of the patient's eye to help visualize where the lesion and leakage are located.
"The total procedure from the time the eye is opened to when it's closed takes about 45 minutes," he adds. "The irradiation actually only lasts about four minutes."
According to Mr. Moses, if and when the product is commercialized, the company plans to separate the handpiece into two parts, so the cannula tip can be disposable. "This will ensure that there's always a sterile environment and reduce the time in the OR required to clean the device," he explains. "The Strontium-90 has a half-life of 29 years, so the non-disposable handpiece would need to be returned to the manufacturing site periodically to be recalibrated and returned, but you'd have many months of uninterrupted use before this would be necessary."
Maximizing Effect and Safety
Mr. Moses says the beneficial effect of the radiation is atrributable to differences between the DNA in healthy and diseased cells that cause the radiation to damage the diseased cells preferentially. "The DNA in diseased cells is not as tightly wound as in normal, healthy cells," he explains. "As a result, the radiation penetrates their DNA more easily, destroying the DNA chains before they can replicate. (Healthy retinal cells are highly resistant to radiation below 60 Gy; current exposure in this protocol is 24 Gy.) "The healthy cells continue regenerating," he adds, "but over time the diseased cells causing the leakage stop regenerating and die off, leaving just the healthy cells in the retinal space. That's why it can take four to six weeks to see the results."
Early research tried two different doses of radiation. "The lower dose in the early study did have an effect, but it wasn't strong enough to arrest the entire lesion," he explains, "so the lesion continued to fester and vision continued to worsen over time." But even with the more potent dose, Mr. Moses says one treatment with the device might not effectively cause the damage required to stop the growth of the unwanted cells if the lesion is larger than 5.5 mm in diameter. "For that reason, as with all other treatments for AMD, it's key for these patients to be seen shortly after the wet lesion presents itself," he says. "When you catch these lesions while they're smaller, the chance of improving or at least maintaining vision is greatly increased."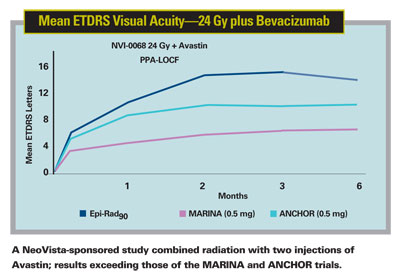
Mr. Moses notes that the device is safe for the patient, surgeon and others in the immediate vicinity because Strontium-90 radiation is easily blocked. "If the tool is activated outside of the eye—which it's not supposed to be—those nearby would receive some radiation exposure," he says. "That's why it's important for everyone present to wear some type of radiation badge or ring that shows the presence of radiation. However, when the device is activated inside the eye, the radiation is completely contained. A Geiger counter next to the eye would pick up no reading."
Although the company has no clinical evidence yet regarding how long the beneficial effect of this procedure ultimately will last, similar treatments using Strontium-90 plaques to treat the wet form of AMD have arrested the disease for several years postop. (Those treatments involved putting the isotope behind the back of the eye, which made it difficult to place correctly, required much longer exposure, and may have compromised extra tissue unnecessarily.) "We believe that using the same isotope, placed directly over the lesion, will allow the treatment to have a longevity of greater than two years," he says. "We recently presented our 12-month data, and lesions continue to be arrested at that time point."
Upping the Ante with Anti-VEGF
One of the most interesting developments involving the Epi-Rad system has been the finding that combining this protocol with an anti-VEGF drug produces even better results. A feasibility study that used an injection of Avastin at the time of irradiation with one follow-up injection a month later found that the combination produced a dramatic improvement in visual acuity—a mean gain of 13.4 letters at six months. (For comparison, the
"A physician at Harvard named Rakesh K. Jain has published a number of papers about using radiation with anti-VEGF drugs to treat cancer," says Mr. Moses. "Some believe that radiation causes an increase in VEGF production, so the combination makes sense. In any case, we know that a tumor, anatomically, shares some traits with a leaking vessel in the back of the eye. Our advisors felt that if it worked so successfully in the back of the eye for tumors, it should also work for active AMD lesions and choroidal neovascularization."
According to Mr. Moses, the next trial sponsored by NeoVista, titled the CNV Secondary to AMD Treated with Beta Radiation Epiretinal Therapy trial, or CABERNET, will compare this protocol (replacing Avastin with Lucentis) to the effectiveness of Lucentis alone; it should begin by the middle of 2007. He notes that CABERNET will have about 40 clinical sites, about 30 of them in the
The company believes that a treatment combining the Epi-Rad system with a drug like Lucentis or Avastin, requiring far fewer injections into the eye than the current recommended Lucentis protocol, should provide significant benefits for both patients and the treating physicians. Clearly, better results with fewer injections would make patients happy and save them money; it would also reduce the workload for the doctor and place less of a burden on the struggling Medicare insurance system.
"We've been hearing a lot about the burden on doctors and practices caused by large numbers of patients coming in frequently for repeat injections of anti-VEGF drugs," says Mr. Moses. "The income generated for the practice from the drug itself is negligible, and the government doesn't reimburse doctors a lot for administering an intraocular injection. The combination treatment eliminates many of the costly, time-and-effort intensive injections, and because it involves vitrectomy may provide better reimbursement for the surgeon's work."
Coming Soon
Mr. Moses says the company hopes to be in a position to submit their PMAs for commercialization of the product in 2009. "We're considering the possibility of launching the product outside of the United States before the study is completed," he says, "but we don't want to take our eye off the ball to start running a commercial operation. We want to make sure the trial is done right."



