Natural Therapies
For the purposes of this discussion, natural therapies include nutritional and herbal supplements as well as treatments from the field of complementary and alternative medicine (CAM), such as acupuncture or apheresis.
In addition to simply being cognizant of what a particular therapy can do to a patient's system, it's also important to know about the use of alternative therapies before any surgical procedures. Some commonly used supplements, like ginkgo, act as anti-coagulants and may increase the risk of bleeding when taken with aspirin, warfarin or another anti-coagulant. Some sources recommend discontinuing all supplement use two to three weeks preop, but according to the American Academy of Ophthalmology, there is inadequate evidence on which to base any recommendations. The AAO website has more information: http://aao.org/education/library/cta/nutr_supp_peri.cfm.
Nutrition and Supplements
Over the last decade, there have been a number of advances in our understanding of the relationship between diet and eye disease. For example, the Nurses' Health Study found regular consumption of spinach, kale and other foods rich in carotenoids to be moderately protective for cataract development in women.3 In another study, women with the healthiest overall diets (fruits, vegetables, and whole grains) were significantly less likely to develop nuclear opacities than those with the least healthy diets.4 The Health Professionals Study found that regular consumption of broccoli and spinach decreased the risk of cataract in men.5 An Italian study found an increased risk of cataract in those with the highest intakes of butter, total fat, salt and oils other than olive oil.6
Specific Nutrients
To evaluate the effect of antioxidant and zinc supplements on the progression of age-related macular degeneration and visual acuity, the well-known Age-Related Eye Disease Study developed a formula of 500 mg vitamin C, 400 IU vitamin E, 15 mg beta-carotene, 80 mg zinc and 2 mg copper in the form of cupric oxide. Researchers found that both zinc alone and antioxidants plus zinc delayed progression to end-stage AMD by 25 percent in those with either intermediate or advanced AMD.7
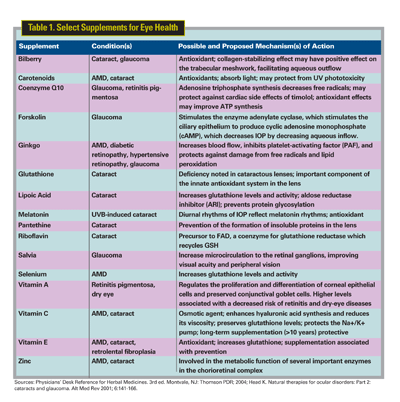 |
The carotenoids lutein and zeaxanthin are pigments in the macula, and their putative protective effect for AMD and cataract may be a result of their ability to absorb light and protect the retina from UV phototoxicity. A recent trial of lutein supplements in patients with AMD showed that both lutein alone and lutein combined with vitamins, minerals and antioxidants improved both objective and subjective visual function.8 Both nutrients appear safe, though their long-term use has not been evaluated.
Some studies have shown cataract formation to be related to deficiencies in glutathione, and cataract patients also tend to be deficient in vitamin A, lutein and zeaxanthin. Nutrients that increase glutathione levels include lipoic acid, vitamins E and C, and selenium. Diabetic cataract, on the other hand, is caused by elevation of polyols catalyzed by the enzyme aldose reductase, and flavonoids are aldose reductase inhibitors (ARIs).9
Pantethine, the active coenzyme form of pantothenic acid (B5), has been demonstrated to inhibit lens opacification in some animal studies.10 Pantethine drops were used in the first clinical trial of an anti-cataract drug in 2000, although the trial was stopped because of drop irritation.11
One of the new drugs, OT-551 (Othera Pharmaceuticals), is metabolized into TP-H, a compound that mimics naturally occurring antioxidants.12 TP-H then acts as a catalytic antioxidant scavenger. OT-551 is in Phase 2 clinical trials as a means of preventing progression of cataracts after vitrectomy. The search continues for the nonsurgical intervention against or prevention of cataracts.
There is evidence that omega-3 supplements (flaxseed oil, evening primrose oil, cod liver oil, etc.) may protect against or be beneficial for dry eye, AMD, diabetic retinopathy and retinitis pigmentosa, among others.
For example, the Women's Health Study found that a higher dietary intake of n-3 fatty acids is associated with a decreased incidence of dry eye in women.13 However, the metabolism, physiological importance, biological effects and pathological role of omega-3 acids remain unexplored. The original AREDS didn't evaluate omega-3 fatty acids, but AREDS II (which is now recruiting) will study both omega-3s and the carotenoids. Some high-risk patients are already taking omega-3 supplements. Patients with clotting disorders or who are taking anti-coagulants should be monitored, and should possibly stop taking the supplements several days preop.
Other Supplements
A number of supplement formulations have been developed for ocular conditions:
• TheraTears Nutrition. This is a blend of flaxseed oil, fish oil and vitamin E, and is currently marketed for dry eye, although to our knowledge no studies have yet been published to demonstrate its effectiveness. Using the controlled adverse environment model, our group has evaluated a novel dry-eye supplement provided by Steven Pratt, MD. The study showed reduction in symptoms after 10 weeks in 71 percent of patients (Pratt SG, et al. IOVS 2005;46:E-Abstract 2047).
• Melatonin. This is a pineal hormone and is a popular aid for sleep and jet lag recovery. Due to its antioxidant properties and its ability to neutralize a variety of free radicals and reactants, melatonin may have a protective effect against cataract, glaucoma and other eye diseases, although research hasn't progressed to interventional studies in humans. However, injections of melatonin have been shown to inhibit UV-induced cataract in rats,14 and oral melatonin reduces IOP in normal patients.15
• Bilberry (Vaccinium myrtillus). Bilberry has a long history of medicinal use for eye conditions. Its active ingredient is the flavonoid anthocyanosides. One clinical study of bilberry extract combined with vitamin E showed arrested cataract formation in 48 of 50 patients with cataract.16 In addition, bilberry improves vision in individuals with diabetic retinopathy17 and has shown promise in a preliminary glaucoma trial.18
• Ginkgo biloba. This supplement, purported to improve memory and mental acuity, also has an effect on ophthalmic artery blood flow.19 A number of human trials have suggested that there exists the potential for using ginkgo in the treatment of AMD and other conditions,20,21 but further research is needed. The flavonol glycosides (quercetin) may be the relevant component in ginkgo extract.
• Forskolin. Twenty-five years ago at Harvard University, Arthur Neufeld, PhD, and others found that forskolin (a triterpene derived from the plant Coleus forskohlii) stimulates adenylate cyclase. This in turn stimulates the ciliary epithelium to produce cyclic adenosine monophosphate (cAMP), which then decreases IOP by decreasing aqueous humor inflow.22,23 While the topical use of forskolin appears promising, at least in animals and humans,24 clinical studies of its use in glaucoma patients are lacking.
• Salvia miltiorrhiza. This herb, also known as Chinese sage or danshen, is an important part of modern Chinese clinical practice. Salvia is an aldose reductase inhibitor that also appears to improve microcirculation of the retinal ganglion cells.25 In one human study, intramuscular injections of salvia improved both visual acuity and peripheral vision in people with mid- to late-stage glaucoma.26
Drugs Derived from Botanicals
The first drugs were natural extracts from plants and other organisms. Today, as many as a quarter of all medications are derived from plant sources, and only an estimated 15 percent of potential plants have been evaluated for medicinal use.27 A few botanical therapies are:
• Systane (Alcon). Systane is a botanically derived therapy for dry-eye patients. It contains the natural gelling agent guar—derived from the guar shrub—which has been modified by the addition of a hydrophobic end group to form hydroxypropyl guar (HP-guar). Systane's formulation promotes a coating that lubricates the normal blinking process and facilitates tissue repair.28 The cross-linkage and gellation of the solution form a matrix that is retained on the ocular surface by hydrophobic bonding to dry areas, as demonstrated by the reduction in corneal staining in clinical trials.29,30
• rEV131 (Evolutec). This is a protein derived from the saliva of arthropods. In pre-clinical models of allergic asthma and allergic conjunctivitis, rEV131 has been shown to bind histamine very tightly and block mast-cell degranulation, reducing itching and inflammation associated with histamine release in the allergic response31 (Chapin M et al. IOVS 2002;43:ARVO E-Abstract 109). Phase 2 trials are planned for 2006.
• Other drugs. There are also several drugs in Phase 2 trials for AMD. Oxigene's combretastatin for cancer and myopic macular degeneration is derived from the root bark of the Cape bush willow tree. Genaera's squalamine, an aminosterol that blocks endothelial cell activation, migration and proliferation, may have uses in AMD and retinopathy.32,33
CAM in Ophthalmology
A variety of CAM treatments for ocular conditions have been assessed by the AAO, and full reports are available on the website: aao.org/education/library/cta/index.cfm. None of the treatments is recommended by the AAO, generally because of a lack of evidence of their effectiveness rather than substantial evidence of risks.
Acupuncture has been used in the treatment of a variety of conditions, including allergy, dry eye and glaucoma. Acupuncture is also used for pain management. One randomized controlled trial demonstrated improvements in dry-eye symptoms with acupuncture compared to placebo.34 However, evidence is lacking to bolster the anecdotal reports.
There are a number of other CAM techniques under investigation for AMD, including apheresis, and microcurrent stimulation.
Natural therapies are increasing in popularity despite a general lack of evidence. Patients don't seem to mind that alternative-therapy practitioners have a fraction of the training of physicians or that supplement makers are as profit-driven as drug makers. Though some natural therapies may someday be proven as effective as drugs, ophthalmologists should be prepared to explain to patients the principles of evidence-based medicine, so that patients are less likely to waste time and money on snake oil.
Dr. Abelson, an associate clinical professor of ophthalmology at Harvard Medical School and senior clinical scientist at Schepens Eye Research Institute, consults in ophthalmic pharmaceuticals. Ms. Lines is a medical writer at Ophthalmic Research Associates in North Andover.
1. Carey B. When trust in doctors erodes, other treatments fill the void. New York Times. February 3, 2006. http://www.nytimes.com/2006/02/03/health/03patient.html. Accessed March 7, 2006.
2. Eisenberg DM, Davis RB, Ettner SL, Appel S, Wilkey S, Van Rompay M, Kessler RC. Trends in alternative medicine use in the United States, 1990-1997: results of a follow-up national survey. JAMA 1998;280:18:1569-75.
3. Chasen-Taber L, Willet WC, Seddon JM, et al. A prospective study of carotenoid and vitamin A intakes and risk of cataract extraction in US women. Am J Clin Nutr 1999;70:509-516.
4. Moeller SM, Jacques PF, Blumberg JB. The potential role of dietary xanthophylls in cataract and age-related macular degeneration. J Am Coll Nutr 2000;19:522S-527S.
5. Brown L, Rimm EB, Giovannucci EL, et al. A prospective study of carotenoid intake and risk of cataract extraction in US men. Am J Clin Nutr 1999;70:517–24.
6. Tavani A, Negri E, La Vecchia C. Food and nutrient intake and risk of cataract. Ann Epidemiol. 1996 Jan;6:1:41-6.
7. Age-Related Eye Disease Study Research Group (AREDS Research Group). A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta-carotene, and zinc for AMD and vision loss: AREDS report no. 8. Arch Ophthalmol. 2001;119:10:1417-36.
8. Richer S, Stiles W, Statkute L, et al. Double-masked, placebo-controlled, randomized trial of lutein and antioxidant supplementation in the intervention of atrophic AMD: the Veterans LAST study. Optometry 2004;75:4:216.
9. Varma SD, Kinoshita JH. Inhibition of lens aldose reductase by flavonoids--their possible role in the prevention of diabetic cataracts. Biochem Pharmacol. 1976;25:22:2505-13.
10. Clark JI, Livesey JC, Steele JE. Delay or inhibition of rat lens opacification using pantethine or WR-77913. Exp Eye Res 1996;62:75-84.
11. Harding JJ. Can drugs or micronutrients prevent cataract? Drugs Aging. 2001;18:7:473-86.
12. Othera Pharmaceuticals. How OT-551 works. Exton, PA: Othera Pharmaceuticals, Inc. http://www.othera.com/producttech_ot551.html.
13. Miljanovic B, Trivedi KA, Dana MR, et al. Relation between dietary n-3 and n-6 fatty acids and clinically diagnosed dry eye syndrome in women. Am J Clin Nutr 2005;82:4:887-93.
14. Bardak Y, Ozerturk Y, Ozguner F, Durmus M, Delibas N. Effect of melatonin against oxidative stress in ultraviolet-B exposed rat lens. Curr Eye Res 2000;20:3:225-30.
15. Samples JR, Krause G, Lewy AJ. Effect of melatonin on IOP. Curr Eye Res 1988;7:7:649-53.
16. Bravetti G. Preventive medical treatment of senile cataract with vitamin E and anthocyanosides: clinical evaluation. Ann Otall Clin Ocul 1989;115:109.
17. Perossini M, et al. Diabetic and hypertensive retinopathy therapy with anthocyanosides: double blind placebo-controlled clinical trial. Ann Ottal Clin Ocul 1987;113:1173-90.
18. Caselli L. Clinical and ERG study on activity of anthocyanosides. Arch Med Int 1985;37:29-35.
19. Chung HS, Harris A, Kristinsson JK, Ciulla TA, Kagemann C, Ritch R. Ginkgo biloba extract increases ocular blood flow velocity. J Ocul Pharmacol Ther 1999;15:3:233-40.
20. Lebuisson DA, Leroy L, Rigal G. [Treatment of senile macular degeneration with Ginkgo biloba extract. A preliminary double-blind drug vs. placebo study] Presse Med. 1986;15:31:1556-8.
21. Fies P, Dienel A. [Ginkgo extract in impaired vision--treatment with special extract EGb 761 of impaired vision due to dry senile macular degeneration] Wien Med Wochenschr 2002;152:15-16:423.
22. Bartels SP, Lee SR, Neufeld AH. Forskolin stimulates cyclic AMP synthesis, lowers intraocular pressure and increases outflow facility in rabbits. Curr Eye Res 1982-83;2:10:673-81.
23. Caprioli J, Sears M. Forskolin lowers intraocular pressure in rabbits, monkeys, and man. Lancet 1983;1:8331:958-60.
24. Meyer BH, Stulting AA, Muller FO, Luus HG, Badian M. The effects of forskolin eye drops on intra-ocular pressure. S Afr Med J 1987;71:9:570-1.
25. Zhu MD, Cai FY. Evidence of compromised circulation in the pathogenesis of optic nerve damage in chronic glaucomatous rabbit. Chin Med J (Engl) 1993;106:12:922-7.
26. Wu ZZ, Jiang YQ, Yi SM, Xia MT. Radix Salviae Miltiorrhizae in middle and late stage glaucoma. Chin Med J (Engl) 1983;96:6:445-7.
27. Natural product pharmaceuticals. The Medical & Healthcare Marketplace Guide, 19th edition; Dorland Healthcare Information, 2004.
28. Ubels JL, Clousing DP, Van Haitsma TA, et al. Pre-clinical investigation of the efficacy of an artificial tear solution containing hydroxypropyl-guar as a gelling agent. Curr Eye Res 2004;28:6:437-44.
29. Christensen MT, Cohen S, Rinehart J, et al. Clinical evaluation of an HP-guar gellable lubricant eye drop for the relief of dryness of the eye. Curr Eye Res 2004 Jan;28:1:55-62.
30. Hartstein I, Khwarg S, Przydryga J. An open-label evaluation of HP-Guar gellable lubricant eye drops for the improvement of dry eye signs and symptoms in a moderate dry eye adult population. Curr Med Res Opin 2005;21:2:255-60.
31. Couillin I, Maillet I, Vargaftig B, et al. Arthropod-derived histamine-binding protein prevents murine allergic asthma. J Immunol 2004;173:5:3281-6.
32. Higgins RD, Yan Y, Geng Y, et al. Regression of retinopathy by squalamine in a mouse model. Pediatr Res 2004;56:1:144-9.
33. Ciulla TA, Criswell MH, Danis RP, Williams JI, McLane MP, Holroyd KJ. Squalamine lactate reduces choroidal neovascularization in a laser-injury model in the rat. Retina 2003;23:6:808-814.
34. Nepp J, Wedrich A, Akramian J, et al. Dry eye treatment with acupuncture: A prospective, randomized, double-masked study. In: Sullivan DA, Dartt DA, Meneray MA, eds. Lacrimal gland, tear film and dry eye syndromes 2. New York: Plenum Press, 1998.



