Over the last several decades, endoscopy-based procedures have revolutionized the field of general surgery, born of the desire to perform less invasive surgery while achieving similar or even improved anatomic and clinical outcomes. Operating times have decreased for surgeons in almost all surgical fields, but perhaps more importantly, patients are now able to achieve equivalent surgical success with less postoperative pain and recovery time. Vitreoretinal surgery is now moving toward less invasive surgery as well, and in this brief article we discuss the pros and cons of 23- and 25-ga. vitrectomy.
Sutureless vitrectomy surgery has become increasingly popular since 2002, when Drs. Gildo Fujii, Eugene de Juan, and Mark Humayun and colleagues first reported their initial experiences using 25-ga. instrumentation.1 Some surgeons have converted to 25-ga. instrumentation for nearly all of their surgical procedures and almost all surgeons have reported at least some experience using 25-gauge vitrectomy. A recent survey by the American Society of Retina Specialists revealed that 80 percent of respondents have tried 25-ga. procedures at least once.2 Despite this trend, many retina specialists are still adhering to "standard" 20-ga. instruments, citing technical difficulties as well as safety concerns for patients. 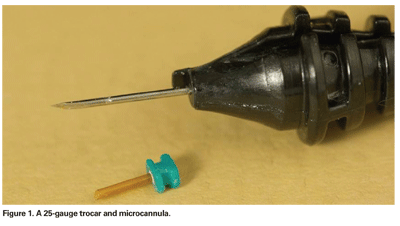
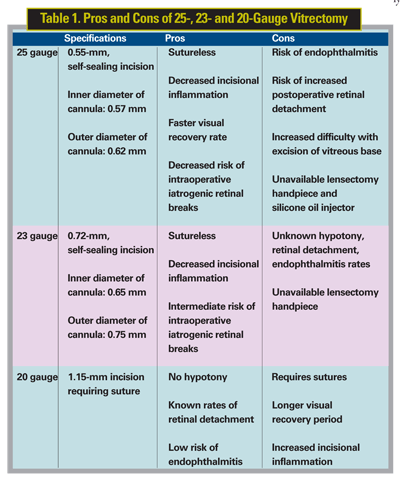
Twenty-five-ga. sutureless vitrectomy involves the insertion of microcannulas (See Figure 1), with an inner and outer diameter of 0.57 and 0.62 mm, respectively, through transconjunctival and scleral incisions, creating a 0.55 mm self-sealing hole. By comparison, sclerotomies via 20-ga. incisions are 1.15 mm in diameter and require sutures for closure. This difference in incision size allows narrow diameter 25-ga. instruments to be used that have, by nature of their smaller gauge, decreased shaft rigidity as compared to their 20-ga. counterparts. Likewise, the 25-ga. small diameter vitrectomy handpiece has a correspondingly smaller aspirating port diameter as compared to 20-ga. instruments (See Figure 2). The difference in port size results in different fluid dynamics during debulking of the vitreous and shaving of the vitreous base. Additionally, and again by nature of their smaller diameter, 25-ga. endoilluminators produce decreased illumination compared to their 20-ga. counterparts.
Proponents of 25-ga. vitrectomy champion these differences as major advances for surgical success and patient comfort. The smaller diameter, sutureless, transconjunctival incisions avoid dissection of the conjunctiva and cautery of the episcleral vessels, which can induce inflammation and scarring. Without the need for sutures, which also induce a local inflammatory response and may result in ocular surface irritation, Lakhanpal and colleagues described reduced intraocular inflammation by postoperative day 30 in 140 patients that underwent 25-ga. procedures as compared to prior studies involving 20-ga. vitrectomy.3 Rizzo and colleagues also described significantly less postoperative intraocular inflammation and discomfort in patients that underwent 25-ga. vitrectomy compared to those that underwent 20-ga. vitrectomy for epiretinal membranes.4 By avoiding conjunctival dissection, small-gauge incisional vitrectomy also allows preservation of filtering blebs and shunts in glaucoma patients. When vitrectomy with 20-ga. instrumentation is performed in patients with filtering blebs, subsequent failure rates may be as high as 70 percent.5 This may be reduced with sutureless vitrectomy procedures. 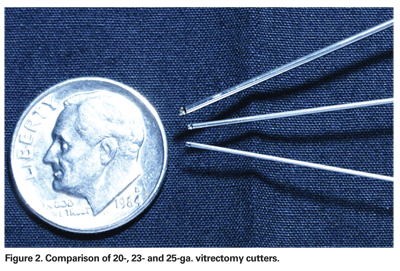
Smaller diameter ports in 25-ga. vitrectomy handpieces result in port-limited flow, theorized to result in less fluid turbulence and a reduced risk of intraoperative iatrogenic retinal breaks. A smaller volume of fluid is aspirated with each open-close cycle in 25-ga. vitrectomy, defined by Steve Charles, MD, as pulse flow.6 With decreased pulse flow, fluidic stability is increased, resulting in port-flow limiting and thereby reducing cutter-induced motion of detached retina, which many feel leads to decreased iatrogenic cutter-induced retinal breaks. Finally, decreased intraoperative times have been reported. Dr. Fujii and colleagues showed that, on average, the total operating time in 25-ga. procedures was 17 minutes, compared to 26 minutes for 20-ga. procedures.1 However, actual vitrectomy times were slightly higher on average in 25-ga. cases compared to 20-ga. cases (15 minutes vs. 14 minutes).
Despite the many supporters of 25-ga. vitrectomy, detractors of sutureless vitrectomy have reservations. By nature of their smaller-gauge diameters, the instruments lack the relative rigidity and stability of 20-ga. instruments. Although technical adjustments such as varying the angle of trocar insertion and increased shaft rigidity of newer generation 25-ga. instruments allow more ease in vitreous base excision, 25-ga. vitrectomy has generally not been favored for vitrectomy for more complicated anterior retinal pathology including phakic rhegmatogenous retinal detachment and proliferative vitreoretinopathy. In patients requiring pars plana lensectomy or silicone oil insertion, 25-ga. instruments are currently unavailable.
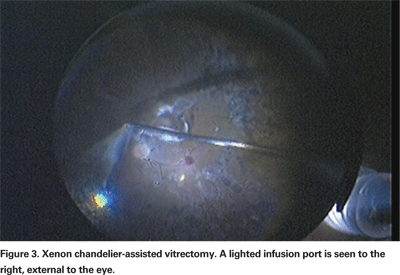
Ongoing improvements in technology may limit concerns with 25-ga. vitrectomy's relatively dim illumination. The power output of standard 25-ga. light probes when initially introduced was approximately 50 to 70 percent less than standard 20-ga. light probes. However, this has largely been addressed with the recent advent of xenon illuminators. As a result, xenon-illuminated 25-ga. endoilluminator probes are now equivalent in brightness to standard 20-ga. endoilluminators. Visualization may also be enhanced with xenon chandeliers, which provide a panoramic view of the fundus and allow surgeons to employ bimanual surgical techniques (See Figure 3).
Postoperative hypotony is an occasionally troublesome complication of sutureless 25-ga. surgery, which may rarely require further suture closure. Fortunately, postoperative hypotony has not consistently been observed in long term studies. Rohit Lakhanpal, MD, and colleagues, however, did report a 3.8 percent incidence of shallow choroidal detachments in 140 patients undergoing 25-ga. vitrectomy, all of which resolved by postoperative day seven.3 Once again, many surgeons have adapted to new 25-ga. technology with a variety of maneuvers, including displacing conjunctiva before making the initial incisions, creating valved scleral tunnels with the trocars, or using partial air-fluid exchange at the end of procedures to seal the wounds internally. Ultimately, if incisions are not self-sealing with sclera, vitreous must play some role in the "sutureless" closure. Vitreous incarceration may increase the risk of retinal detachment or endophthalmitis. David Williams, MD, presented data at the 2006 AAO Subspecialty Day meeting that showed increased retinal detachment, endophthalmitis and hypotony rates when comparing 25-ga. and 20-ga. vitrectomy.7 The retinal detachment rate in 356 cases when using 25-ga. vitrectomy was 3.4 percent, but only 0.6 percent in 337 cases using 20-ga. vitrectomy. He also reported three cases (0.8 percent) of endophthalmitis and two cases (0.6 percent) of hypotony in the 25-ga. group, but none in either category in the 20-ga. group. Additional long-term studies are needed to fully assess the risk-benefit ratio of 25-ga. vitrectomy.
With lack of instrument rigidity, relatively poorer lighting, and less than ideal flow rates having established themselves as the main concerns of 25-ga. vitrectomy, a new sutureless vitrectomy system was introduced that directly addressed these issues: 23-ga. vitrectomy. The first 23-ga. vitrectomy system to be commercially available was introduced by Dutch Ophthalmic Research Center (DORC), and recently Alcon has introduced its new 23-ga. system as well. Claus Eckhardt, MD, was the first to describe the use of 23-ga. vitrectomy in 2005, citing improved cutting rates and shaft rigidity similar to conventional 20-ga. vitrectomy, but with the advantages of sutureless 25-ga. instrumentation.8 The inner and outer diameters of the 23-ga. microcannula are 0.65 and 0.75 mm respectively; it is inserted through a 0.72- mm "single-step" incision made by an angled blade. Instead of a perpendicular incision with a 25-ga. trocar, a tangential incision at a 30 to 45-degree angle is made through the sclera, allowing for a valved or beveled self-sealing wound after the cannula is removed. While 25-ga. cannulas are made from flexible polyamide, 23-ga. cannulas are made from metal, which are much more firmly embedded within the sclera. The diameters of the cutter and instruments are larger relative to 25-ga. instruments, resulting in higher illumination and flow rates (See Figures 2 and 4). Although this relatively small increase in port diameter appears insignificant, Poiseuille's law states that flow rates are equal to the fourth power of the radius of a cylindrical tube. Thus, small changes in the diameter of the vitreous cutter, for example, lead to very large changes in flow rates. Though only about 0.1-mm greater in diameter than the 25-ga. cutter, the 23-ga. cutter's flow rates are significantly higher, leading to more efficient debulking of vitreous. 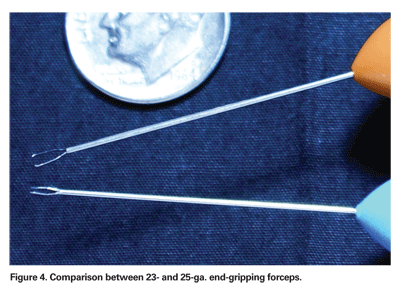
Additionally, critical maneuvers such as inducing a mechanical posterior vitreous detachment are easier with higher suction and flow rates. The rigidity of the instruments themselves allow the surgeon to much more easily manipulate and excise peripheral vitreous. Finally, increasing the diameter of the endoillimunator probe also allows for greater illumination and therefore visualization. However, given the relatively recent introduction of 23-ga. instruments, long-term follow-up on the risk of endophthalmitis, hypotony, postoperative choroidal effusion and retinal detachment remains unknown. "Hybrid" vitrectomy, in which different gauge incisions and instruments are applied during the course of a single operation, is already being described by innovative surgeons. These procedures involve the use of sutureless infusion cannulas and larger incisions for instruments when retinal pathology dictates.
Robert Machemer, the inventor of modern vitrectomy more than two decades ago, might scarcely recognize the procedure he invented. However, he would likely be proud of the ongoing evolution of our understanding of fluid dynamics and other contributing factors to efficient and safe vitrectomy. Smaller-gauge instruments represent these ongoing improvements in technology. Nevertheless, patient selection and the surgeon's comfort with smaller-gauge sutureless vitrectomy will likely dictate the overall adoption of its use in vitrectomy.
Supported in part by the San Francisco Retinal Foundation and the Pacific Vision Foundation. Dr. Roe practices in the Department of Ophthalmology at
1. Fujii GY, de Juan E, Jr., Humayun MS, et al. A new 25-gauge instrument system for transconjunctival sutureless vitrectomy surgery. Ophthalmology 2002;109:1807-12; discussion 13.
2. Mittra R. Preferences and Trends Survey. American Society Retina Specialists, 2006.
3. Lakhanpal RR, Humayun MS, de Juan E, Jr., et al. Outcomes of 140 consecutive cases of 25-gauge transconjunctival surgery for posterior segment disease. Ophthalmology 2005;112:817-24.
4. Rizzo S, Genovesi-Ebert F, Murri S, et al. 25-gauge, sutureless vitrectomy and standard 20-gauge pars plana vitrectomy in idiopathic epiretinal membrane surgery: A comparative pilot study. Graefe's Arch Clin Exp Ophthalmol 2006;244:472-9.
5. Thompson WS, Smiddy WE, Flynn HW, Jr.,
6. Charles S. An engineering approach to vitreoretinal surgery. Retina 2004;24:435-44.
7. Williams DF. Complications of 25 Gauge Vitrectomy.
8. Eckardt C. Transconjunctival sutureless 23-gauge vitrectomy. Retina 2005;25:208-11.



