First-line Standard: The PGAs
“Like most of my colleagues, I tend to use a prostaglandin as my first-line option,” says Jason Bacharach, MD, medical director and founding partner of North Bay Eye Associates in Sonoma County, Calif. Tony Realini, MD, MPH, professor of ophthalmology and director of the glaucoma service, clinical research and the glaucoma fellowship at West Virginia University Eye Institute, says that for his newly diagnosed patients who don’t elect primary SLT, generic latanoprost is his first choice. “The prostaglandin class of drugs provides the optimal efficacy, safety and convenience thanks to once-daily dosing—and lower cost with the generic formulation of latanoprost.” Latanoprost also figures prominently in a recently released line of preservative-free compounded formulations called Simple Drops (Imprimis), ranging from the drug alone, to fixed-dose combinations of multiple drugs. The company says the combinations may increase patient compliance while reducing costs.
The prostaglandin analog class of drugs also includes bimatoprost (Lumigan; Allergan), travoprost (Travatan Z; Alcon), and tafluprost, which is available in a preservative-free formulation (Zioptan; Akorn Pharmaceuticals). As monotherapy, latanoprost, bimatoprost, and travoprost have all been shown to have comparable IOP-lowering efficacy.1 The PGAs have virtually no systemic side effects, but possible ocular side effects include ocular/conjunctival hyperemia, eyelash growth, darkening of the iris, darkening of the periorbital skin and loss of periorbital fat.
 |
| Topical prostaglandin analog drops are widely accepted as first-line therapeutic agents for newly diagnosed glaucoma and ocular hypertension patients. New drugs and drug-delivery systems will soon vie for positions in the treatment paradigm, however. |
For many patients, however, the advantages of the PGAs outweigh their disadvantages. They are generally the first-line therapy of choice before beta-blockers such as timolol; alpha-adrenergic agonists such as brimondine; carbonic anhydrase inhibitors such as dorzolomide; and the cholinergenic agonist pilocarpine.
Latanoprostene Bunod
“The reality is that only one drug so far has been better than latanoprost,” says Dr. Realini. “That’s latanoprostene bunod (LBN), and it’s just a little bit better.”
Latanoprostene bunod, a nitric oxide-donating prostaglandin F2 alpha analog, incorporates two mechanisms of action into one novel agent. Administered once daily, LBN is quickly metabolized into latanoprost acid and a nitric oxide-donating moiety called butanediol mononitrate. This new drug is thought to increase uveoscleral outflow via the latanoprost acid while also increasing the outflow of aqueous through the trabecular meshwork. “Greater IOP control with LBN is probably attributable to the nitric oxide moiety that‘s attached to the latanoprost,” Dr. Realini explains. “When it’s dosed, it breaks apart into standard latanoprost and nitric oxide. Latanoprost improves uveoscleral outflow, while nitric oxide works within the smooth muscle of the trabecular meshwork to increase trabecular outflow. These two complementary mechanisms provide an additive IOP reduction.”
LBN 0.024% outperformed latanoprost 0.005% at lowering IOP in the Phase II Voyager study, which compared the safety and efficacy of LBN at various concentrations to latanoprost 0.005%. After 28 days, a 0.024% concentration of LBN reduced IOP by 9 mmHg in patients with a mean diurnal baseline IOP of 26.01 mmHg, representing 1.23 mmHg greater IOP reduction than in patients randomized to the latanoprost group.2 A small number of study patients reported eye pain at the drop instillation site in both groups, with more LBN patients than latanoprost patients experiencing this. In Phase III studies, once-daily LBN demonstrated efficacy and safety, and was noninferior to3 or better than4 twice-daily timolol at lowering IOP in patients with open-angle glaucoma or ocular hypertension. Valeant, its subsidiary Bausch + Lomb and Nicox have announced a PDUFA date of August 24, 2017: The new drug’s trade name is Vyzulta.
Despite LBN’s superior IOP-lowering in comparison to latanoprost in trials, Dr. Realini says there’s no sure way to predict the effect it will have on glaucoma practice patterns. “It’s too early to tell how much of the market it will capture,” he notes. “Based on what we know about these drugs so far, there may be an efficacy advantage to LBN over latanoprost, but I’m not sure whether that translates to being cost effective in the long run.”
Three Mechanisms: Netarsudil
There are four ways that an IOP-lowering drug can do its job: increasing outflow through the trabecular meshwork; increasing uveoscleral outflow; decreasing episcleral venous pressure; and decreasing aqueous humor production. Netarsudil, a rho kinase (ROCK) and norepinephrine transporter (NET) inhibitor, is thought to target three of these. Dr. Bacharach, who has been an investigator for Rhopressa (Aerie Pharmaceuticals) says, “Besides increasing outflow from the trabecular meshwork, it appears from preclinical trials that it also lowers episcleral venous pressure. Interestingly, it also appears to reduce fluid production in the eye.”
In the ROCKET 4 study of Rhopressa,5 a trial required for the European regulatory approval process but not for the manufacturer’s NDA to FDA, Rhopressa lowered IOP in eyes with a mean baseline IOP ranging from 20.7 to 22.4 mmHg to 16.3 to 17.8 mmHg at three months. Hyperemia occurred in 42.2 percent of study eyes, with most of it mild to moderate. Aerie has announced a PDUFA date of February 28, 2018.
Dr. Bacharach looks forward to integrating Rhopressa into his armamentarium primarily as a second-line therapy. “If you look at the Phase II study6 [of which Dr. Bacharach is an author], you’ll see that at lower starting pressures, i.e., below 24 mmHg, this drug worked equivalently and was non-inferior to latanoprost,” he says. “It might be that in people who have low pressures there’s an opportunity for this drug to be the first-line therapy because it’s also dosed once daily. But in general, its most common position will likely be as a first-line adjunct to a prostaglandin.
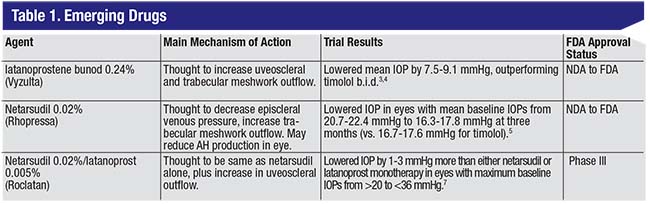 |
| A list of novel IOP-lowering drugs currently in the FDA pipeline. These agents all target more than one mechanism of action to lower intraocular pressure in patients with glaucoma or ocular hypertension. |
“It’s really complementary to the prostaglandins and appears to have an additive effect,” he continues. “It’s been demonstrated that patients who were on prior prostaglandin therapy, even with a washout, seem to get an additive effect when Rhopressa is added to the eye. The bottom line is that they’re synergistic and they probably have complementary mechanisms. So theoretically, when you add Rhopressa to a PGA, you’ve affected every way there is to lower intraocular pressure. The uveoscleral outflow is addressed by the PGA: You’ve affected the other three mechanisms through netarsudil,” he says.
A fixed-combination agent, Roclatan (Aerie) is behind Rhopressa in the FDA-approval process. Roclatan aims to target all four known mechanisms of IOP-lowering by harnessing the apparent additive effect of netarsudil and latanoprost. Roclatan has been demonstrated to have superior IOP-lowering abilities to either of its active components alone.7 Dr. Bacharach believes that Roclatan has great potential as a first-line drug, based on its performance in trials.
Rhopressa represents a milestone in IOP-lowering therapeutics, according to Dr. Bacharach. “There hasn’t been a new class of medicine in two decades,” he says. “This class of medicines, and this drug in particular, is poised to be our first new option in a very long time. I use a variety of drugs as second-line options, and I think this drug will really launch to the top of my list for additive therapy.”
All in the Delivery?
Even the safest and most effective IOP-lowering eye drop is devoid of therapeutic value if the script goes unfilled or the bottle sits in a medicine cabinet. Topical PGAs remain a therapeutic mainstay in the fight against elevated IOP, and many manufacturers are developing novel delivery systems to overcome the biggest and best-known barrier to efficacy: poor compliance. “The prostaglandins are very effective and very safe, so it’s going to be hard to beat them,” says Dr. Realini. “One of the ways in which we might beat them is by finding a delivery system that allows us to reduce the dosing frequency and move the responsibility for dosing from the patient to the physician.”
Ahmad A. Aref, MD, assistant professor and residency director at Illinois Eye and Ear Infirmary in Chicago, believes that the available and emerging IOP-lowering drugs leave little room for improvement: Instead, getting medicine to consistently reside in the eye is his current priority in glaucoma therapy. “I think that we’ve probably reached very close to our limit in terms of what we’re going to be able to achieve with new medicated eye drops,” he says. “I’m excited to hear about the new classes of medicines that are coming, but I would say that where things are really ripe for innovation is the question of how those drugs are going to be delivered,” he says.
Dr. Aref notes that topical drops are too often an all-or-nothing proposition. “One thing that I’ve started to do in my office, if I’m questioning why someone’s glaucoma doesn’t seem to be under good control, is ask the patient to put an eye drop in at the office where I can watch them,” he says. “Many patients are elderly and lack the physical ability to instill an eye drop in their eye; it can actually be a little scary to watch them attempting it in my office—not to mention multiple times per day at home without any assistance.”
Dr. Aref, who recently reviewed emerging sustained-delivery systems for glaucoma drugs,8 says that devices that don’t penetrate the ocular surface may have an edge in terms of their safety profile. Devices in development include, but aren’t limited to, a bimatoprost-eluting silicone-coated periocular ring (ForSight VISION5, Menlo Park, Calif./Allergan) made to sit between the upper and lower fornices of the eye; canalicular plugs that deliver travoprost (Ocular Therapeutix; Bedford, Mass.) and latanoprost-delivering punctal plugs (Mati Therapeutics; Austin, Texas). The above three technologies are currently in clinical trials.
“The bimatoprost ring and the punctal plug are both implanted on the outside of the eye and are very, very low risk since there’s no incision; there’s essentially no risk of infection on the inside of the eye. They’re very safe,” Dr. Aref says. The ring is available in an assortment of diameters and can deliver medication continuously for six months, although decreasing amounts of drug are released into the tear film over time. In Phase II testing, both the ring and topical timolol achieved IOP reduction of greater than 20 percent compared to baseline.9 Although the ring didn’t fully demonstrate noninferiority to timolol drops in that study, Dr. Aref points out that candidates for the ring would likely be patients who are unsuitable for drops due to poor adherence, making the rings a viable alternative. There were no unexpected adverse events, and subjects reported minor effects that commonly occur with bimatoprost use. The ring was retained in most study eyes for the six-month period and dislodgement was easily detected.
Also in the pipeline are sustained-release anterior chamber implants that deliver bimatoprost (Bimatoprost SR; Allergan) and travoprost (Travoprost XR; Envisia Therapeutics and iDose; Glaukos). The Bimatoprost SR, a biodegradable device currently in Phase III trials, demonstrated comparable IOP lowering in treated eyes at four different doses to fellow eyes treated with bimatoprost drops at 16 weeks and six months in data from a 24-month Phase I/II trial.10 Transient adverse events occurred within two days of the injection in some implanted eyes, but no eyes needed the device explanted and there were no serious adverse events. Later-onset hyperemia was less common in implanted eyes than in the fellow eyes treated with bimatoprost drops.
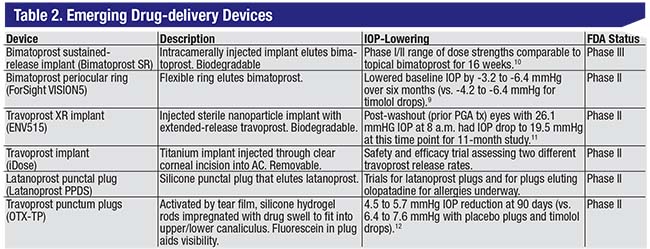 |
| A list of sustained-release drug-delivery devices in clinical trials. Though none of the emerging technologies listed here delivers new medications, they may prove innovative aids to compliance for those who cannot successfully follow a topical eye-drop regimen. |
Like the Bimatoprost SR, the Travoprost XR implant is biodegradable; the iDose is titanium and meant to be retrieved after it runs out of active travoprost.
“The injection-type therapy, i.e. the bimatoprost injection or the travoprost injection, seems to be more efficacious and last for a longer period of time,” says Dr. Aref. “There’s also no risk of dislodging it, compared to the ring or the punctal plug, which can be fairly easily dislodged. They don’t seem to be as effective as medications given in an eye-drop form, though, so I think it’s truly a judgment call in terms of risk versus benefit at this stage with the data that we have.”
Dr. Aref notes that the injectable devices sidestep the issue of potential ocular surface damage from long-term glaucoma eye drop use. “We’re now recognizing that very frequent exposure of the ocular surface to the preservatives in these eye drops can be very harmful over the long term, and may potentially even affect the outcome of glaucoma surgery, if a patient eventually needs it. Something like these injected implants would circumvent the potential exposure of the eye to preservatives, which can be very harmful,” he says.
Dr. Aref also predicts that intracamerally injected IOP-lowering therapeutics will gain acceptance in light of the growth of injections to treat AMD. “I do think that patients with glaucoma are going to accept injection therapy for their disease because they likely have friends who get injections for macular degeneration, which have gotten a lot of publicity,” he says.
Dr. Realini and Dr. Aref both wonder whether extended-release technologies will have an obvious target market, however. “In a patient who’s well-controlled on a topical prostaglandin and who is not getting worse, is there any advantage to moving from a generic drop once a day to an expensive device?” wonders Dr. Realini. “What if they don’t work quite as well as eye drops? Would the convenience factor be worth losing a point or two of IOP control? In some patients it might be. It’s good to have options, but when we acquire them all at once, it’s hard to know what to do with them,” he observes.
Dr. Aref adds, “If somebody is fully adherent with their eye-drop regimen and not having any adverse effects, it’s going to be very difficult to make a case for any of these devices.” He does think that insurers might pay more up front to avoid the fiscal ramifications of uncontrolled glaucoma in patients who’ve clearly failed to benefit from IOP-lowering drops. “Sustained drug delivery is worth considering for someone who acknowledges difficulty with adherence or has barriers to adherence, whether they are financial or physical limitations to being able to use eye drops. That could capture a significant percentage of patients,” he says.
A Puzzle with Added Pieces
“The drugs that are in development and may be poised for approval in the coming months offer some advantages over our current first-line therapy of generic latanoprost,” says Dr. Realini. “These are innovative products. They aren’t yet another beta-blocker or PGA. But each of these drugs or drug-delivery systems also has limitations. It’s not clear at this point that the balance of benefits and disadvantages will position any of these new products higher on the treatment spectrum than latanoprost.”
Although it remains unclear where new drugs and devices will fit into the glaucoma treatment paradigm, Dr. Aref says that change is a given. “The way we traditionally think about treating glaucoma is all going to change,” he states. “I think that physicians just need to be accepting of that, and be excited about it, because glaucoma is a field that really needs innovation. There’s just so much more that our patients are going to need and expect over time.” REVIEW
Dr. Bacharach is an investigator for Aerie Pharmaceuticals. Dr. Realini is a consultant for Alcon, Novartis, Inotek, New World Medical and Bausch + Lomb Dr. Aref has received a speaking honorarium from Novartis and is a consultant for New World Medical.
1. Parrish RK, Palmberg P, Sheu WP. The XLT Study Group: A comparison of latanoprost, bimatoprost, and travoprost in patients with elevated intraocular pressure: A 12-week, randomized, masked-evaluator multicenter study. Am J Ophthalmol 2003;135:3:688-703.
2. Weinreb RN, Ong T, Scassellati Sforzolini B, et al. A randomised, controlled comparison of latanoprostene bunod and latanoprost 0.005% in the treatment of ocular hypertension and open angle glaucoma: The VOYAGER study. Br J Ophthalmol 2015;99:6:738-45.
3. Medeiros FA, Martin KR, Peace J, Sforzolini BS, et al. Comparison of latanoprostene bunod 0.024% and timolol maleate 0.5% in open-angle glaucoma or ocular hypertension: The LUNAR Study. Am J Ophthalmol 2016;168:250-9.
4. Weinreb RN, Scassellati Sforzolini B, Vittitow J, Liebmann J. Latanoprostene bunod 0.024% vs. timolol maleate 0.5% in subjects with open-angle glaucoma or ocular hypertension: The APOLLO study. Ophthalmology 2016;123:5:965-73.
5. Khouri AS, Heah T, Kopczynski C, et al. A double-masked, randomized, parallel study of Netarsudil Ophthalmic Solution 0.02% QD compared to timolol maleate ophthalmic solution, 0.5% BID in patients with elevated intraocular pressure (ROCKET-4). Paper presented at ARVO 2017 Annual Meeting. May 9 2017. Baltimore, MD. E-Abstract 2461.
6. Bacharach J, DuBiner HB, Levy B, et al. Double-masked, randomized, dose-response study of AR-13324 vs. Latanoprost in patients with elevated intraocular pressure. Ophthalmol 2015;122:2:302-307.
7. Aerie Pharmaceuticals company communication. Phase 3 topline results of Mercury 2 study. NCT20674854. http://files.shareholder.com/downloads/AMDA-29CD43/4355053862x0x944296/95EE87E6-B885-463C-8D82-26A52ACBDD04/AERIE_Topline_Mercury_2_Final.pdf [accessed May 24, 2017].
8. Aref A. Sustained drug delivery for glaucoma: Current data and future trends. Cur Opin Ophthalmol 2017;28:2:169-174.
9. Brandt JD, Sall K, DuBiner H, Benza R, Alster Y, Walker G, et al. Six-month intraocular pressure reduction with a topical bimatoprost ocular insert: Results of a Phase II randomized controlled study. Ophthalmology 2016;123:8:1685-1694.
10. Lewis RA, Christie WC, Day DG, et al. Bimatoprost sustained-release implants for glaucoma therapy: 6-month results from a Phase I/II clinical trial. Amer J Ophthalmol 2017;175:137–147.
11. Envisia Therapeutics. Envisia therapeutics releases interim ENV515 (Travoprost XR) Phase 2 data demonstrating 11-month duration-of-action after a single dose in patients with glaucoma. Press Release. 3 February 2017. http://www.envisiatherapeutics.com/wp-content/uploads/2017/02/Draft-Press-Release-ENV515-Cohort-2-11-Month-Data-Release-2-2-2017-FINAL-FINAL.pdf [Accessed May 18, 2017].
12. Ocular Therapeutix, Inc. Ocular Therapeutix TM reports on topline results of phase 2b glaucoma clinical trial. Press Release. 22 October 2015. http://investors.ocutx.com/phoenix.zhtml?c=253650&p=irol-newsArticle&ID=2100516 [Accessed May 18, 2017].




