Each year at ARVO, refractive researchers push the boundaries of their understanding outward a little more, studying new techniques for the first time and going over older techniques in order to hone their skills. At this year's meeting, surgeons' studies cover everything from the femtosecond laser to pain control after PRK. Here's the latest.
(Unless stated otherwise, the researchers have no financial interest in the techniques or technology discussed in their abstracts.)
Making Flaps
Researchers from
The physicians took 1,000 consecutive LASIK cases performed using the Intralase and the WaveLight Eye-Q laser and compared them to a matched group of 1,000 cases using the Eye-Q laser and a microkeratome, the Moria M2. Of the 1,000 femtosecond LASIK cases, 825 eyes were myopic and 175 eyes were hyperopic. The same surgeon performed all the surgeries.
The mean preoperative uncorrected vision for the myopic eyes was 20/80, and their mean postoperative uncorrected vision was 20/17, with a mean flap thickness of 105 ±5 µm. For the hyperopic eyes that underwent femtosecond LASIK, the mean preop uncorrected vision was 20/60, and the mean postop vision was 20/15 with an average flap thickness of 132 ±7 µm. Similar visual acuity results were obtained in the microkeratome LASIK group.
Three minor flap complications occurred in the femtosecond LASIK group. Two were managed intraoperatively, and one was treated within two months by a microkeratome without loss of uncorrected vision. In the microkeratome group, there were six flap complications, which all required subsequent PRK. In the femtosecond cases, there were two cases of an epithelial abrasion and buttonhole, but no incidences of flap slippage, epithelial downgrowth or diffuse lamellar keratitis. In the microkeratome group, there were six cases of flap striae, 22 cases of epithelial downgrowth, three buttonholes, 65 cases of epithelial abrasions and 12 cases of DLK.547
Researchers at the Mayo Clinic in
In a paired-eye study, 15 patients had each of their eyes randomized to flap creation with the IntraLase or the Bausch & Lomb Hansatome. The researchers examined the corneas with confocal microscopy before surgery and at three years postop. They measured central keratocyte density in the anterior and posterior halves of the stroma in the flap by using a custom program that objectively identified bright objects (presumed to be keratocyte nuclei) on confocal images. They compared cell density between treatments by using paired t-tests (unless otherwise stated), and calculated the minimum detectable difference (MDD) for non-significant differences (p=0.05, ß=0.20).
In the anterior half of the stromal flap, mean keratocyte density before bladeless LASIK (43,071 ±7,421 cells/mm3) didn't differ from keratocyte density before LASIK with the microkeratome (39,228 ±5,371 cells/ mm3; p=0.13; MDD=7,200 cells/ mm3). At three years, keratocyte density in the anterior stromal flap also didn't differ between treatments (bladeless: 34,740 ±7,359 cells/mm3; microkeratome: 35,693 ±6,040 cells/ mm3; p=0.72, MDD=7,800 cells/ mm3).
In the posterior half of the stromal flap, keratocyte density before IntraLase LASIK (27,269 ±5,081 cells/ mm3) did not differ from keratocyte density before LASIK with the microkeratome (27,269 ±4591 cells/mm3; p=0.99, MDD=5,350 cells/mm3).
At three years, keratocyte density in the posterior stromal flap also didn't differ between treatments (bladeless: 21,340 ±4,592 cells/mm3; microkeratome: 20,813 ±4,631 cells/mm3; p=0.71, MDD=4,200 cells/mm3). Keratocyte density at three years was 19 percent lower in the anterior flap (p=0.005) and 22 percent lower in the posterior flap (p<0.001) compared to preop densities after IntraLase LASIK. It was 9 percent lower in the anterior flap (p=0.01, signed-rank test) and 24 percent lower in the posterior flap (p<0.001) after LASIK with the microkeratome.4524
A prospective, randomized study from researchers at the
The researchers evaluated 190 consecutive eyes of 95 bilateral IntraLase LASIK patients in a randomized contralateral-eye study. They analyzed the following: superior and temporal hinge position; 45-degree and 90-degree hinge angle; and 100 µm and 130 µm corneal flap thicknesses. Preop, they also evaluated central corneal Cochet-Bonnet esthesiometry, an ocular surface disease index questionnaire, Schirmer's test with anesthesia, tear breakup time and corneal fluorescein and conjunctival lissamine green staining. They then evaluated these factors at postop week one and one, three, six and 12 months postoperatively.
The physicians report that corneal sensation was reduced at all time points after LASIK with improvement over 12 months (p<0.001). There was no difference in corneal sensation between the different hinge positions, angles or flap thicknesses at any time point. Overall, ocular surface disease index score was increased at one week (p<0.0001), one month (p<0.0001, and three months postop, (p=0.046). The percentage of patients with tear breakup time greater than 10 seconds was significantly lower at one week and one month (p<0.0001), and mean corneal fluorescein staining was significantly higher at one week (p<0.0001). There was no difference in ocular surface index score, Schirmer's, tear breakup time or lissamine green and fluorescein staining in contralateral eyes when comparing hinge position, hinge angle or flap thickness.560
Ablation Outcomes
A collaborative effort of surgeons from
The retrospective study consisted of 58 eyes of 31 patients with primary hyperopia or hyperopic astigmatism. The IR software wouldn't engage in 36 of the eyes. Both groups had comparable baseline characteristics, with a preop spherical equivalent of +1.69 ±0.84 D in the IR group and +1.81 in the no-IR group (p=0.876).
At the six-month follow-up visit, 91 percent of the IR group and 86 percent of the no-IR group had the same best-corrected vision as they did preop or had gained one or two lines of vision. No eye in either group lost more than one line of best-corrected vision (p=1.00). Sixty-eight percent of the IR patients and 72 percent of the no-IR group had the same uncorrected vision as preop or had gained one to two lines of uncorrected vision.
No eye lost more than one line of uncorrected vision (p=1.00). Enhancements were necessary in 9 percent of the IR group and 4 percent of the no-IR group. Overall, the researchers say there were no statistically significant differences between the groups.552
Surface Ablation
Researchers from the
In their first study, the physicians prospectively performed Epi-LASIK on 298 eyes and PRK on 290, all for moderate amounts of myopia or myopic astigmatism. The mean preop spherical equivalent refraction in Epi-LASIK was -2.97 ±1.19 D (r: -1 to -6.25 D). For PRK, it was -2.94 ±1.06 D (r: -1 to -6 D) (p=0.819).
Postop uncorrected acuity was as good or better than preop acuity in 83.6 percent of Epi-LASIK eyes compared to 71.7 percent of PRK patients at six months (p=0.027).
Uncorrected vision of 20/15 or better was more likely following Epi-LASIK compared to PRK at one month (26.2 percent vs. 17.8 percent), three months (63.5 percent vs. 49.3 percent), six months (77.8 percent vs. 57.9 percent) and 12 months (76.2 percent vs. 61.9 percent). Low-contrast visual acuity (5 and 25 percent) was comparable between the treatment groups at all time points except 5 percent LCVA at 12 months, which was unchanged or better than preop in 93.9 percent of epi-LASIK patients vs. 78.3 percent of PRK patients. There was no difference between the two procedures in best-corrected acuity, predictability or clinically significant corneal haze at any time point.
The researchers say that, though Epi-LASIK resulted in equivalent or superior visual outcomes compared to PRK, it remains to be seen whether the procedure is a suitable surface-ablation alternative in patients with higher degrees of myopia.553
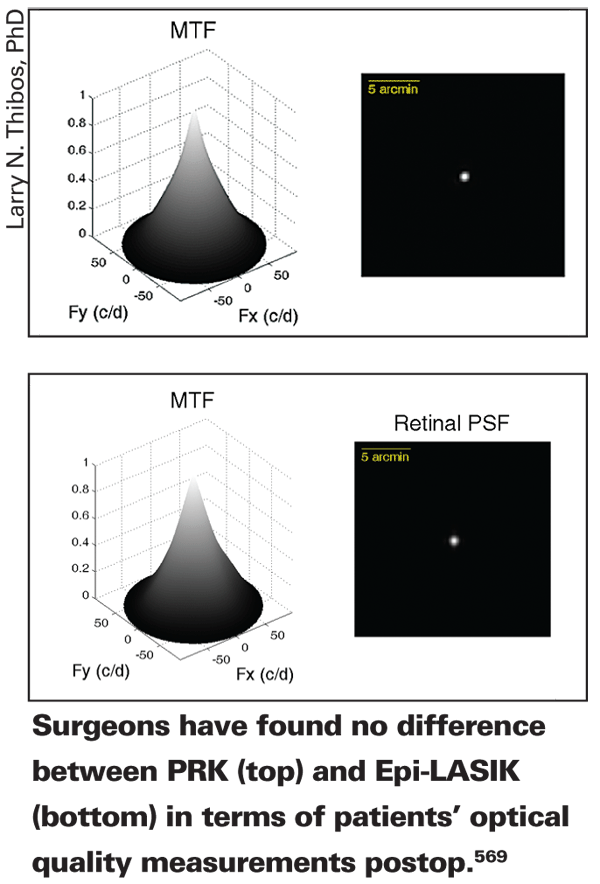
In another study, the same researchers found no difference between PRK and Epi-LASIK in terms of average postop sphere and optical quality. In this prospective study, the physicians compared outcomes from 287 PRK cases to 165 Epi-LASIK surgeries. The procedures were performed with conventional Alcon LADARVision 6000 ablations. The surgeons converted postop sphero-cylindrical refractions into power vectors consisting of mean sphere (M), regular astigmatism (J0) and oblique astigmatism (J45). They measured monochromatic aberrations up to the 4th order with a Shack-Hartmann wavefront analyzer and determined total absolute higher-order aberrations RMS. Using Fourier optics and a subject's wavefront, the researchers calculated the area of a 2D radially averaged modulation transfer function for two pupil sizes.
For mean sphere (Epi-LASIK: M=+0.12; PRK: M=-0.04), regular astigmatism (Epi-LASIK: J0=+0.07; PRK: J0=+0.03), and oblique astigmatism (Epi-LASIK: J45=-0.011; PRK: J45=+0.013), there were no statistically significant differences across procedures. Total absolute HOA RMS didn't differ significantly for either a 3-mm pupil (Epi-LASIK: RMS=0.098; PRK: RMS=0.092), or a 6-mm pupil (Epi-LASIK: RMS=0.88; PRK: RMS=0.80). The area underneath a normalized 2D MTF was almost identical across procedures and pupil diameter (3-mm pupil [p=0.25], 6-mm pupil [p=0.84]) and didn't differ significantly. The physicians conclude that, given the additional cost and training required for Epi-LASIK, the procedure offers no additional benefit over PRK in terms of optical quality or higher order aberrations.569
Researchers at Walter Reed also performed a prospective, randomized study on 40 patients undergoing PRK for myopia or myopic astigmatism. The aim of the study was to compare the efficacy of oral gabapentin (n=20) to placebo (n=20) for the control of postop pain.

The surgeons debrided the epithelium with the Amoils epithelial scrubber (Innovative Excimer Solutions Inc.) and performed the ablations with the Allegretto Wave Eye-Q laser (WaveLight/Alcon). They used standard medications following PRK. Patients were randomized to receive gabapentin 300 mg or placebo t.i.d., beginning two days prior to surgery and continuing for four days after surgery, for a total of seven days. The primary outcome measures were pain levels reported at two hours postop, on postop days one through four and maximum pain levels daily over a 24-hour period, using the visual analog pain scale. Secondary outcome measures were frequency of use of an oral narcotic pain-reliever, Percocet (oxycodone 5 mg/acetaminophen 325 mg), assessed by performing a pill count of the remaining Percocet tablets on the day-four office visit. A Mann-Whitney test compared the two groups' Percocet use.
Among the two treatment groups, there were no statistically significant differences in current or maximum reported pain at any time point or decrease in frequency of use of Percocet for breakthrough pain following PRK. Gabapentin showed no improvement in postoperative pain measures compared to placebo.590
Imaging
Researchers at
In a prospective study, 50 normal subjects (100 eyes) underwent sequential measurements of their CCT by Pentacam, Visante anterior segment optical coherence tomography, Orbscan II and ultrasound pachymetry at the hands of an experienced examiner. The average age of the subjects was 38 ±12.1 years (r: 21 to 60 years).
The average CCT by the various methods was as follows: Pentacam, 553.7 ±37.0 µm; AS-OCT, 525.2 ±38.3 µm; Orbscan II, 559.2 ±40.6 µm and ultrasound, 541.4 ±39.1 µm. The researchers say that measurements between the four modalities were strongly correlated (Pearson correlation r value >0.95). There was a statistically significant difference between the measurements obtained in CCT by all four methods (p<0.0001). Compared to ultrasound, Pentacam and Orbscan II significantly overestimated CCT; the former by an average of 12.33 ±1.81 µm, and the latter by 17.80 ±2.37 µm. OCT underestimated it by 16.16 ±1.79 µm.5098
Implants
At this year's meeting, surgeons from
The consecutive case series includes 85 eyes that underwent ICL (Model V4) implantation. The main outcome measures were central vaulting, best-corrected acuity and complication rates. The average preoperative spherical equivalent refraction was -16.31 ±5.43 D (r: -7.75 to -31.75 D) and the average preoperative best-corrected vision was 0.52 ± 0.24 (a little better than 20/40).
The average follow-up in the study was 74.1 ±23.1 months (r: 26 to 124 months). At one month postop, the refraction was reduced to -0.97 ±1.95 D and best-corrected vision increased to 0.71 ±0.93 (slightly better than 20/30). The refraction has remained stable throughout the study period, the physicians say. Though they didn't provide the actual numbers, the researchers say that mean endothelial cell density showed an early reduction after implantation followed by a continuous slow decline. Central lens vaulting decreased from 456 ±215 µm after a week to 187 ±172 µm after 10 years. Patients with higher lens rises had lower central vaulting, and low central vaulting was related to cataract development. The surgeons had to remove ICLs due to secondary cataract development in 16.2 percent of the cases after 10 years.5615

Researchers from
In 390 healthy Caucasian subjects with an average age of 41 ±16 years (r: 10 to 80), researchers used the Orbscan II-system to determine the chamber depth. They assessed the influence of age, gender and spherical equivalent, and then quantified the impact of these factors using linear regression. The study patients' mean spherical equivalent refraction was 1.3 ±2 D.
After measurements, the anterior chamber depth was 2.87 ±0.04 mm in male subjects and 2.81±0.37 mm in female patients, differences that weren't statistically significant (p>0.12).
The decline of the anterior depth occurred in a linear fashion with 0.013 mm of decrease per year, reaching 0.65 mm in 50 years. The study doctors report that the negative correlation between age and anterior chamber depth was statistically significant (r=-0.54; p<0.05). For a risk assessment of phakic intraocular lens implantations, the researchers used model calculations, and found a significant negative correlation between the anterior chamber depth and the spherical equivalent (r=-0.42; p<0.05).6120
Austrian researchers receiving support from Acufocus say that their initial study of the company's ACI 7000 presbyopic corneal inlay found that it has the potential for good efficacy.
In this small, prospective study, the surgeons compared nine eyes of nine patients before and six months after implantation of the ACI, a 5 to 10 µm thick, microperforated artificial aperture designed to restore some near visual acuity. They used a femtosecond laser to create a corneal pocket at a depth of 320 µm and then implanted the device in the pocket. The patients had an average age of 55 (r: 48 to 60 years).
The surgeons measured uncorrected visual acuity using the Optec 6500P Vision Tester, and the accommodative range with a double-pass analyzer called the Optical Quality Analysis System II (Visiometrics;
The researchers observed a mean accommodative range of 0.9 +0.4 D preoperatively (r: 0.5 to 1.5 D), which improved six months after surgery to 2.6 +0.7 D (r: 1.25 to 3.5 D). Every patient improved within this period, the physicians report. Over the same time period, the uncorrected near vision as measured on the ETDRS Near Vision Chart changed from a Snellen equivalent of 20/63 preoperatively to 20/32 six months postoperatively.
The researchers say that, though the results in this small number of patients appear to be good, further study in more patients will be necessary to draw any definitive conclusions.2802
Dr. Probst is national medical director for TLC Laser Centers.